- Materials
-
- 5–10 ml whole blood
- Consumables
-
- 10X phosphate-buffered saline (PBS), pH 7.4 (Thermo Fisher, 70011044)
- Percoll, 1.135 g/ml (Cytiva, 17-0891-01)
- (Optional) dimethyl sulfoxide (DMSO) (Sigma-Aldrich, 20-139)
- Foetal bovine serum (FBS) (Gibco™, A3840401)
- (Optional) chilled foetal bovine serum (FBS) (Gibco™, A3840401)
- 50 ml centrifuge tubes
- 2 ml Eppendorf DNA LoBind tubes
- Equipment
-
- Pasteur pipettes
- Temperature-controlled centrifuge with rotor for 2 ml and 50 ml tubes
- Ice bucket with ice
- P1000 pipette and tips
- P200 pipette and tips
- P100 pipette and tips
- P20 pipette and tips
- P10 pipette and tips
- Optional equipment
-
- Liquid nitrogen and canister
- -80°C freezer storage
-
PBMC sample preparation for Pore-C DNA extraction
Before starting the Pore-C DNA extraction, the peripheral blood mononuclear cells (PBMCs) must be isolated from whole blood whilst maintaining cell viability. Approximately 5–10 ml of whole blood should yield sufficient PBMCs for the Pore-C DNA extraction. If necessary, combine multiple aliquots of whole blood to achieve a final 5–10 ml pooled sample. Using the below method, approximately 10 million PBMCs are prepared in aliquots of 1X PBMCs supplemented with 2% FBS. Approximately 10 million PBMCs are taken forwards into the day 1 of the Pore-C experiment.
Users may isolate PBMCs by any means they feel are most appropriate for the whole blood sample to be used, provided that:
- PBMCs are isolated as soon as possible from fresh blood and no later than 24 hours.
- PBMC are isolated using a method optimised for cell viability.
- The whole blood is not mixed with any additives, except for anticoagulants (e.g. K2-EDTA), which are acceptable and will not interfere with the Pore-C DNA extraction.
If users have a minimum of approximately 10 million PBMCs which have been isolated previously which satisfy these requirements, they may start sample extraction directly from day 1 of the Pore-C experiment.
-
Prepare three solutions in preparation for white blood cells isolation:
- 500 ml of 1X PBS supplemented with 2% FBS final concentration and store at room temperature.
Reagent Volume 10X PBS 50 ml Foetal bovine serum (FBS) 10 ml Nuclease-free water 440 ml Total 500 ml - 100 ml of 1X PBS supplemented with 60% Percoll final concentration and store at room temperature.
Reagent Volume 10X PBS 10 ml Percoll 60 ml Nuclease-free water 30 ml Total 100 ml - (Optional for storage) 2 ml of FBS supplemented with 20% DMSO final and store at 4°C.
Reagent Volume FBS 1,600 µl DMSO 400 µl Total 2,000 µl -
Allow the whole blood sample to warm to room temperature and then dilute with equal volume of room temperature 1X PBS supplemented with 2% FBS. Transfer the diluted blood to a 50 ml centrifuge tube.
-
Centrifuge at 800 x g at 20°C for 10 minutes with the brake off to prevent remixing of the separated fractions.
-
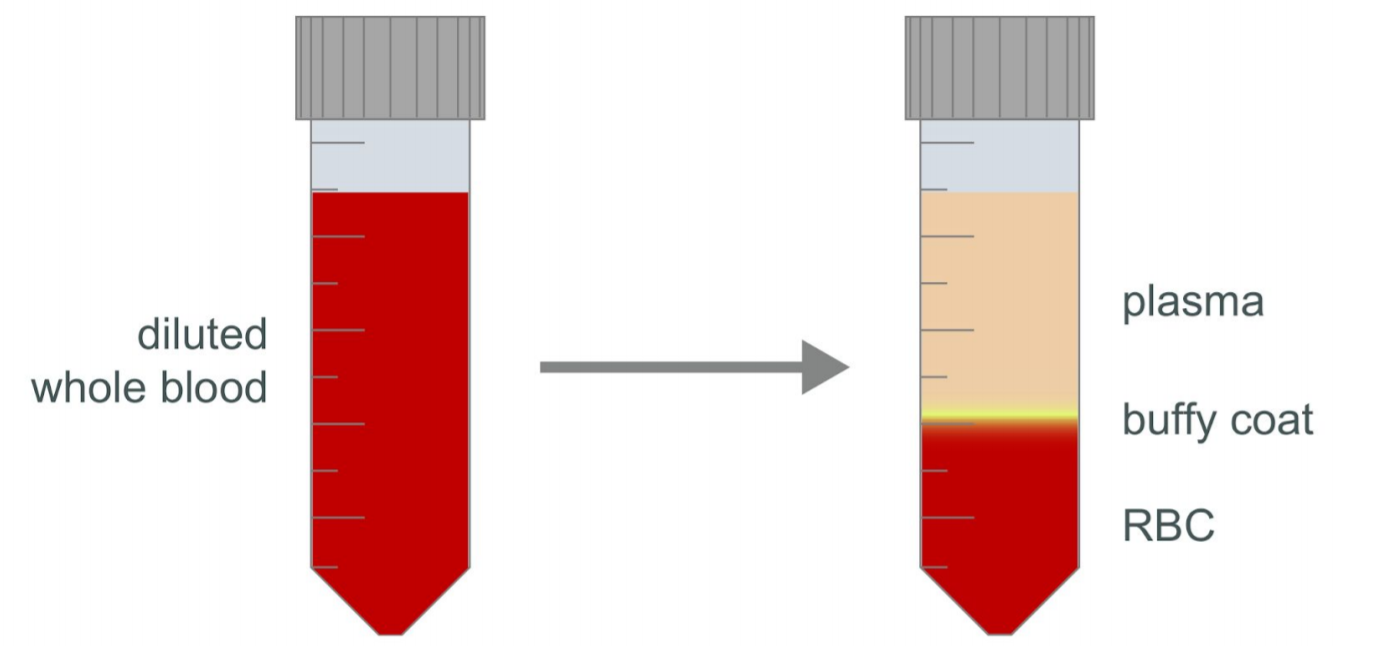
After centrifugation, the whole blood should have separated into the plasma, buffy coat and red blood cells. Check the turbidity of the plasma layer (the top layer). If it is not clear, centrifuge at 800 x g at 20°C for a further 10 minutes with the brake off.
-
Using a Pasteur pipette, remove as much of the plasma layer as possible without disturbing the layer of buffy coat. Gently remove the buffy coat layer, taking care to draw as little of the red blood cell layer as possible. Transfer the recovered buffy coat to a fresh 50 ml centrifuge tube.
-
Make up the recovered buffy coat sample to 25 ml of 1X PBS supplemented with 2% FBS.
-
Aliquot 20 ml of 1X PBS supplemented with 60% Percol in a fresh 50 ml centrifuge tube.
-
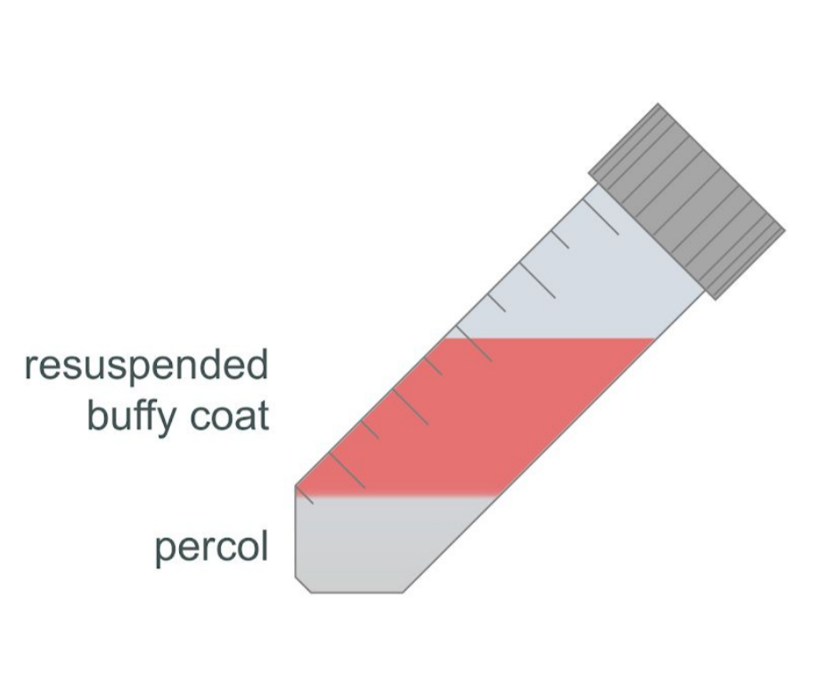
Using a fresh Pasteur pipette, very gently layer the diluted buffy coat sample over the Percol layer at a 45° angle.
-
Centrifuge at 350 x g at 20°C for 40 minutes with slow acceleration and with the brake off.
-
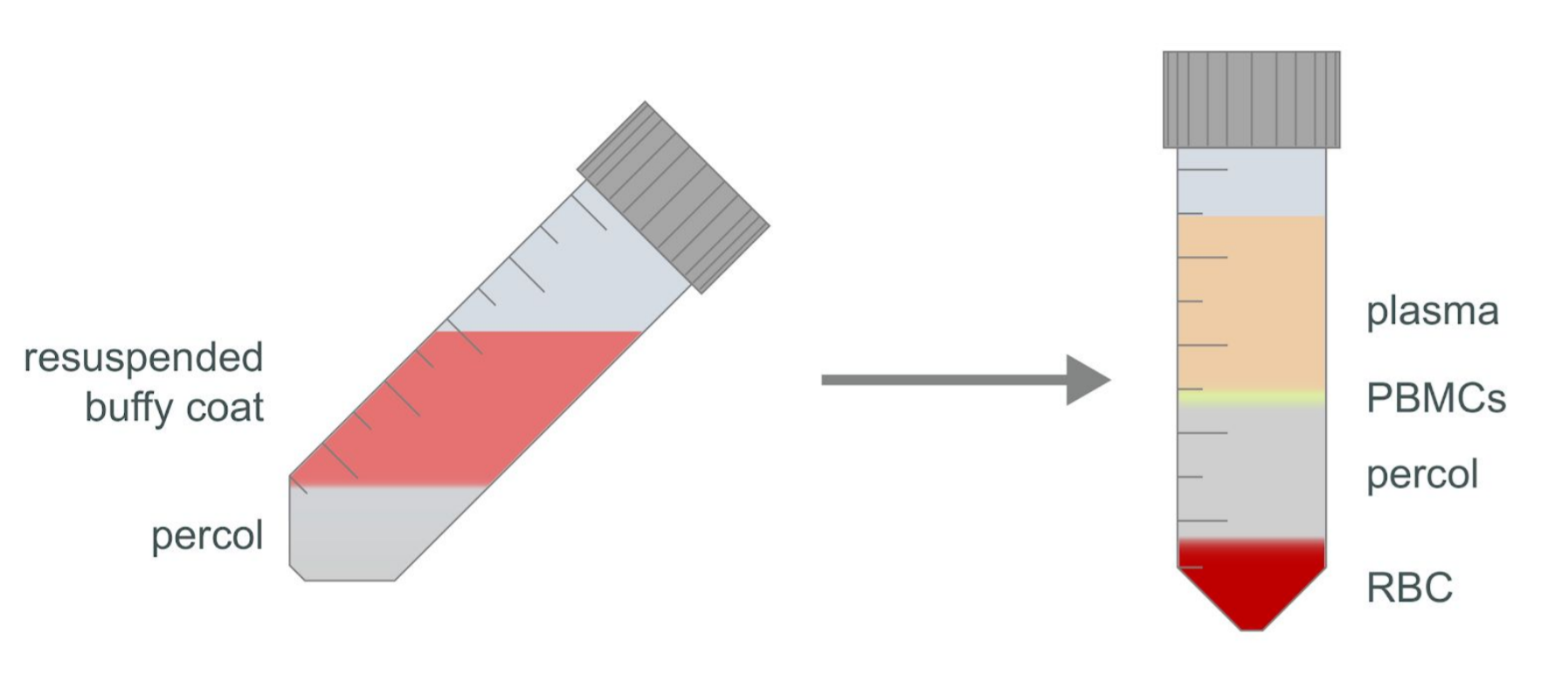
Check the turbidity of the plasma layer and the formation of the white blood cells layer. If the plasma layer is not clear or the PBMC layer is not well defined, continue to centrifuge at 350 x g at 20°C for a further 20 minutes using slow acceleration with the brake off.
-
Using a Pasteur pipette, remove as much of the plasma layer as possible without disturbing the layer of white blood cells, then gently remove the layer of PBMCs. It is acceptable to draw plasma with the layer of white blood cells; however, take care to draw as little of the Percol layer as possible.
-
Transfer the recovered white blood cells to a fresh 50 ml centrifuge tube.
-
Resuspend the recovered white blood cells in 50 ml of room temperature 1X PBS supplemented with 2% FBS.
-
Centrifuge at 350 x g at 20°C for 15 minutes with the brake on.
-
Aspirate and discard the supernatant. Gently resuspend the white blood cells in 25 ml of room temperature 1X PBS supplemented with 2% FBS. Centrifuge at 350 x g at 20°C for 15 minutes with the brake on.
-
Repeat the previous step.
-
Aspirate and discard the supernatant. Gently resuspend the white blood cells in another 25 ml of room temperature 1X PBS supplemented with 2% FBS.
-
Centrifuge at 200 x g at 20°C for 10 minutes with the brake on.
-
Assuming every 1 ml of whole blood originally used will yield approximately 1.5 million white blood cells, resuspend cells to approximately 10 million white blood cells/ml in room temperature 1X PBS supplemented with 2% FBS.
-
Transfer an aliquot of approximately 10 million white blood cells total to a fresh 2 ml Eppendorf DNA LoBind tube.
-
Cool on ice for 5 minutes.
-
Optional actionThe cells can be stored if Pore-C sample extraction cannot be started immediately.
- Centrifuge at 350 x g at 4°C for 2 minutes with the brake on.
- Aspirate and discard the supernatant, then resuspend the white blood cells pellet in 1 ml of chilled FBS.
- Once resuspended, slowly mix in 1 ml of chilled FBS supplemented with 20% DMSO, drop by drop.
Note: As DMSO is mixed with water, energy is released as heat. Adding DMSO to the white blood cells suspension drop by drop prevents heat shock to the cells. - Snap freeze aliquots of white blood cells in liquid nitrogen then store at –80°C.


