- Materials
-
- Input RNA in 10 mM Tris-HCl, pH 8.0
- Native Barcoding Expansion 96 (EXP-NBD196)
- Native Barcoding Expansion 1-12 (EXP-NBD104) and 13-24 (EXP-NBD114) if multiplexing 24 samples
- Flow Cell Priming Kit (EXP-FLP002)
- SFB Expansion (EXP-SFB001)
- Sequencing Auxiliary Vials (EXP-AUX001)
- Adapter Mix II Expansion (EXP-AMII001)
- Consumables
-
- LunaScript™ RT SuperMix Kit (NEB, cat # E3010)
- Q5® Hot Start High-Fidelity 2X Master Mix (NEB, cat # M0494)
- SARS-CoV-2 primers (lab-ready at 100 µM, IDT)
- Nuclease-free water (e.g. ThermoFisher, cat # AM9937)
- Agencourt AMPure XP beads (Beckman Coulter™, A63881)
- Freshly prepared 80% ethanol in nuclease-free water
- Qubit dsDNA HS Assay Kit (ThermoFisher, cat # Q32851)
- NEB Blunt/TA Ligase Master Mix (NEB, M0367)
- NEBNext® Ultra II End Repair / dA-tailing Module (NEB, E7546)
- NEBNext Quick Ligation Module (NEB, E6056)
- DNA 12000 Kit & Reagents - optional (Agilent Technologies)
- 1.5 ml Eppendorf DNA LoBind tubes
- 5 ml Eppendorf DNA LoBind tubes
- Eppendorf twin.tec® PCR plate 96 LoBind, semi-skirted (Eppendorf™, cat # 0030129504) with heat seals
- Equipment
-
- Hula mixer (gentle rotator mixer)
- Magnetic rack, suitable for 1.5 ml Eppendorf tubes
- Centrifuge capable of taking 96-well plates
- Microfuge
- Vortex mixer
- Thermal cycler
- Multichannel pipettes suitable for dispensing 0.5–10 μl, 2–20 μl and 20–200 μl, and tips
- P1000 pipette and tips
- P200 pipette and tips
- Ice bucket with ice
- Timer
- Optional equipment
-
- Agilent Bioanalyzer (or equivalent)
- Qubit fluorometer (or equivalent for QC check)
- Eppendorf 5424 centrifuge (or equivalent)
- PCR hood with UV steriliser (optional but recommended to reduce cross-contamination)
- PCR-Cooler (Eppendorf)
- Stepper pipette and tips
-
Input RNA guidelines
Where sample RNA is added to the below reaction, it is likely advantageous to follow the dilution guidelines proposed by Josh Quick:
qPCR Ct Dilution factor 18–35 none 15–18 1:10 12–15 1:100 If the sample has a low copy number (ct 18–35), use up to 16 µl of sample. Use nuclease-free water to make up any remaining volume. Take note to be aware that co-extracted compounds may inhibit reverse transcription and PCR.
-
Native Barcoding Expansion 96 (EXP-NBD196) contents
Kits in batches NBD196.10.0007 onwards have barcodes ordered in columns on the plate:
Kits in batches prior to NBD196.10.0007 have barcodes ordered in rows:
Name Acronym Cap colour No. of vials Fill volume per vial (μl) Native Barcode 01-96 NB01-96 - 1 plate 40 μl per well Adapter Mix II AMII Green 1 70 -
Native Barcoding Expansion 1-12 (EXP-NBD104) and 13-24 (EXP-NBD114) contents
EXP-NBD104 kit contents
Name Acronym Cap colour No. of vials Fill volume per vial (μl) Native Barcode 01-12 NB01-12 White 12 20 Adapter Mix II AMII Green 1 40
EXP-NBD114 kit contents
Name Acronym Cap colour No. of vials Fill volume per vial (μl) Native Barcode 13-24 NB13-24 White 12 20 Adapter Mix II AMII Green 1 40 -
Flow Cell Priming Kit contents (EXP-FLP002)
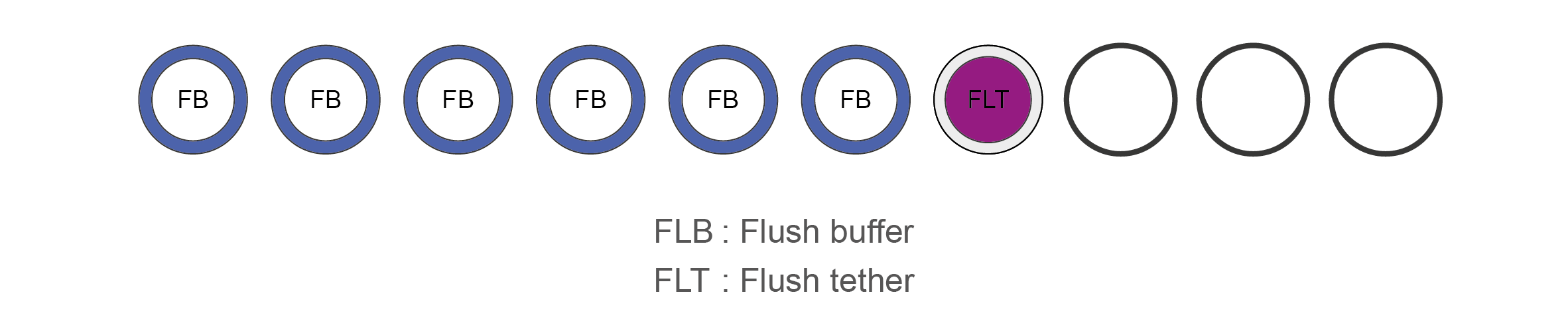
Name Acronym Cap colour No. of vials Fill volume per vial (μl) Flush Buffer FB Blue 6 1,170 Flush Tether FLT Purple 1 200 -
SFB Expansion contents (EXP-SFB001)
Name Acronym Cap colour No. of vials Fill volume per vial (μl) Short Fragment Buffer SFB Grey 4 1,800 -
Sequencing Auxiliary Vials contents (EXP-AUX001)
Name Acronym Cap colour No. of vials Fill volume per vial (μl) Sequencing Buffer SQB Red 6 300 Elution Buffer EB Black 2 200 Loading Beads LB Pink 2 360 -
Adapter Mix II Expansion contents (EXP-AMII001)
Name Acronym Cap colour No. of tubes Fill volume per vial (μl) Adapter Mix II AMII Green 2 40 -
Adapter Mix II Expansion use
Protocols that use the Native Barcoding Expansions require 5 μl of AMII per reaction. Native Barcoding Expansions EXP-NBD104/NBD114 and EXP-NBD196 contain sufficient AMII for 6 and 12 reactions, respectively (or 12 and 24 reactions when sequencing on Flongle). This assumes that all barcodes are used in one sequencing run.
The Adapter Mix II expansion provides additional AMII for customers who are running subsets of barcodes, and allows a further 12 reactions (24 on Flongle).
-
Native barcode sequences
Below is the full list of our native barcode (NB01-96) sequences. The first 24 unique barcodes are available in the Native Barcoding Kit 24 V14 (SQK-NBD114.24). The Native Barcoding Kit 96 V14 (SQK-NBD114.96) include the first 24 native barcodes, with the additional 72 unique barcodes. The native barcodes are shipped at 640 nM.
In addition to the barcodes, there are also flanking sequences which add an extra level of context during analysis.
Barcode flanking sequences:
Forward sequence: 5' - AAGGTTAA - barcode - CAGCACCT - 3'
Reverse sequence: 5' - GGTGCTG - barcode - TTAACCTTAGCAAT - 3'Native barcode sequences
Component Forward sequence Reverse sequence NB01 CACAAAGACACCGACAACTTTCTT AAGAAAGTTGTCGGTGTCTTTGTG NB02 ACAGACGACTACAAACGGAATCGA TCGATTCCGTTTGTAGTCGTCTGT NB03 CCTGGTAACTGGGACACAAGACTC GAGTCTTGTGTCCCAGTTACCAGG NB04 TAGGGAAACACGATAGAATCCGAA TTCGGATTCTATCGTGTTTCCCTA NB05 AAGGTTACACAAACCCTGGACAAG CTTGTCCAGGGTTTGTGTAACCTT NB06 GACTACTTTCTGCCTTTGCGAGAA TTCTCGCAAAGGCAGAAAGTAGTC NB07 AAGGATTCATTCCCACGGTAACAC GTGTTACCGTGGGAATGAATCCTT NB08 ACGTAACTTGGTTTGTTCCCTGAA TTCAGGGAACAAACCAAGTTACGT NB09 AACCAAGACTCGCTGTGCCTAGTT AACTAGGCACAGCGAGTCTTGGTT NB10 GAGAGGACAAAGGTTTCAACGCTT AAGCGTTGAAACCTTTGTCCTCTC NB11 TCCATTCCCTCCGATAGATGAAAC GTTTCATCTATCGGAGGGAATGGA NB12 TCCGATTCTGCTTCTTTCTACCTG CAGGTAGAAAGAAGCAGAATCGGA NB13 AGAACGACTTCCATACTCGTGTGA TCACACGAGTATGGAAGTCGTTCT NB14 AACGAGTCTCTTGGGACCCATAGA TCTATGGGTCCCAAGAGACTCGTT NB15 AGGTCTACCTCGCTAACACCACTG CAGTGGTGTTAGCGAGGTAGACCT NB16 CGTCAACTGACAGTGGTTCGTACT AGTACGAACCACTGTCAGTTGACG NB17 ACCCTCCAGGAAAGTACCTCTGAT ATCAGAGGTACTTTCCTGGAGGGT NB18 CCAAACCCAACAACCTAGATAGGC GCCTATCTAGGTTGTTGGGTTTGG NB19 GTTCCTCGTGCAGTGTCAAGAGAT ATCTCTTGACACTGCACGAGGAAC NB20 TTGCGTCCTGTTACGAGAACTCAT ATGAGTTCTCGTAACAGGACGCAA NB21 GAGCCTCTCATTGTCCGTTCTCTA TAGAGAACGGACAATGAGAGGCTC NB22 ACCACTGCCATGTATCAAAGTACG CGTACTTTGATACATGGCAGTGGT NB23 CTTACTACCCAGTGAACCTCCTCG CGAGGAGGTTCACTGGGTAGTAAG NB24 GCATAGTTCTGCATGATGGGTTAG CTAACCCATCATGCAGAACTATGC NB25 GTAAGTTGGGTATGCAACGCAATG CATTGCGTTGCATACCCAACTTAC NB26 CATACAGCGACTACGCATTCTCAT ATGAGAATGCGTAGTCGCTGTATG NB27 CGACGGTTAGATTCACCTCTTACA TGTAAGAGGTGAATCTAACCGTCG NB28 TGAAACCTAAGAAGGCACCGTATC GATACGGTGCCTTCTTAGGTTTCA NB29 CTAGACACCTTGGGTTGACAGACC GGTCTGTCAACCCAAGGTGTCTAG NB30 TCAGTGAGGATCTACTTCGACCCA TGGGTCGAAGTAGATCCTCACTGA NB31 TGCGTACAGCAATCAGTTACATTG CAATGTAACTGATTGCTGTACGCA NB32 CCAGTAGAAGTCCGACAACGTCAT ATGACGTTGTCGGACTTCTACTGG NB33 CAGACTTGGTACGGTTGGGTAACT AGTTACCCAACCGTACCAAGTCTG NB34 GGACGAAGAACTCAAGTCAAAGGC GCCTTTGACTTGAGTTCTTCGTCC NB35 CTACTTACGAAGCTGAGGGACTGC GCAGTCCCTCAGCTTCGTAAGTAG NB36 ATGTCCCAGTTAGAGGAGGAAACA TGTTTCCTCCTCTAACTGGGACAT NB37 GCTTGCGATTGATGCTTAGTATCA TGATACTAAGCATCAATCGCAAGC NB38 ACCACAGGAGGACGATACAGAGAA TTCTCTGTATCGTCCTCCTGTGGT NB39 CCACAGTGTCAACTAGAGCCTCTC GAGAGGCTCTAGTTGACACTGTGG NB40 TAGTTTGGATGACCAAGGATAGCC GGCTATCCTTGGTCATCCAAACTA NB41 GGAGTTCGTCCAGAGAAGTACACG CGTGTACTTCTCTGGACGAACTCC NB42 CTACGTGTAAGGCATACCTGCCAG CTGGCAGGTATGCCTTACACGTAG NB43 CTTTCGTTGTTGACTCGACGGTAG CTACCGTCGAGTCAACAACGAAAG NB44 AGTAGAAAGGGTTCCTTCCCACTC GAGTGGGAAGGAACCCTTTCTACT NB45 GATCCAACAGAGATGCCTTCAGTG CACTGAAGGCATCTCTGTTGGATC NB46 GCTGTGTTCCACTTCATTCTCCTG CAGGAGAATGAAGTGGAACACAGC NB47 GTGCAACTTTCCCACAGGTAGTTC GAACTACCTGTGGGAAAGTTGCAC NB48 CATCTGGAACGTGGTACACCTGTA TACAGGTGTACCACGTTCCAGATG NB49 ACTGGTGCAGCTTTGAACATCTAG CTAGATGTTCAAAGCTGCACCAGT NB50 ATGGACTTTGGTAACTTCCTGCGT ACGCAGGAAGTTACCAAAGTCCAT NB51 GTTGAATGAGCCTACTGGGTCCTC GAGGACCCAGTAGGCTCATTCAAC NB52 TGAGAGACAAGATTGTTCGTGGAC GTCCACGAACAATCTTGTCTCTCA NB53 AGATTCAGACCGTCTCATGCAAAG CTTTGCATGAGACGGTCTGAATCT NB54 CAAGAGCTTTGACTAAGGAGCATG CATGCTCCTTAGTCAAAGCTCTTG NB55 TGGAAGATGAGACCCTGATCTACG CGTAGATCAGGGTCTCATCTTCCA NB56 TCACTACTCAACAGGTGGCATGAA TTCATGCCACCTGTTGAGTAGTGA NB57 GCTAGGTCAATCTCCTTCGGAAGT ACTTCCGAAGGAGATTGACCTAGC NB58 CAGGTTACTCCTCCGTGAGTCTGA TCAGACTCACGGAGGAGTAACCTG NB59 TCAATCAAGAAGGGAAAGCAAGGT ACCTTGCTTTCCCTTCTTGATTGA NB60 CATGTTCAACCAAGGCTTCTATGG CCATAGAAGCCTTGGTTGAACATG NB61 AGAGGGTACTATGTGCCTCAGCAC GTGCTGAGGCACATAGTACCCTCT NB62 CACCCACACTTACTTCAGGACGTA TACGTCCTGAAGTAAGTGTGGGTG NB63 TTCTGAAGTTCCTGGGTCTTGAAC GTTCAAGACCCAGGAACTTCAGAA NB64 GACAGACACCGTTCATCGACTTTC GAAAGTCGATGAACGGTGTCTGTC NB65 TTCTCAGTCTTCCTCCAGACAAGG CCTTGTCTGGAGGAAGACTGAGAA NB66 CCGATCCTTGTGGCTTCTAACTTC GAAGTTAGAAGCCACAAGGATCGG NB67 GTTTGTCATACTCGTGTGCTCACC GGTGAGCACACGAGTATGACAAAC NB68 GAATCTAAGCAAACACGAAGGTGG CCACCTTCGTGTTTGCTTAGATTC NB69 TACAGTCCGAGCCTCATGTGATCT AGATCACATGAGGCTCGGACTGTA NB70 ACCGAGATCCTACGAATGGAGTGT ACACTCCATTCGTAGGATCTCGGT NB71 CCTGGGAGCATCAGGTAGTAACAG CTGTTACTACCTGATGCTCCCAGG NB72 TAGCTGACTGTCTTCCATACCGAC GTCGGTATGGAAGACAGTCAGCTA NB73 AAGAAACAGGATGACAGAACCCTC GAGGGTTCTGTCATCCTGTTTCTT NB74 TACAAGCATCCCAACACTTCCACT AGTGGAAGTGTTGGGATGCTTGTA NB75 GACCATTGTGATGAACCCTGTTGT ACAACAGGGTTCATCACAATGGTC NB76 ATGCTTGTTACATCAACCCTGGAC GTCCAGGGTTGATGTAACAAGCAT NB77 CGACCTGTTTCTCAGGGATACAAC GTTGTATCCCTGAGAAACAGGTCG NB78 AACAACCGAACCTTTGAATCAGAA TTCTGATTCAAAGGTTCGGTTGTT NB79 TCTCGGAGATAGTTCTCACTGCTG CAGCAGTGAGAACTATCTCCGAGA NB80 CGGATGAACATAGGATAGCGATTC GAATCGCTATCCTATGTTCATCCG NB81 CCTCATCTTGTGAAGTTGTTTCGG CCGAAACAACTTCACAAGATGAGG NB82 ACGGTATGTCGAGTTCCAGGACTA TAGTCCTGGAACTCGACATACCGT NB83 TGGCTTGATCTAGGTAAGGTCGAA TTCGACCTTACCTAGATCAAGCCA NB84 GTAGTGGACCTAGAACCTGTGCCA TGGCACAGGTTCTAGGTCCACTAC NB85 AACGGAGGAGTTAGTTGGATGATC GATCATCCAACTAACTCCTCCGTT NB86 AGGTGATCCCAACAAGCGTAAGTA TACTTACGCTTGTTGGGATCACCT NB87 TACATGCTCCTGTTGTTAGGGAGG CCTCCCTAACAACAGGAGCATGTA NB88 TCTTCTACTACCGATCCGAAGCAG CTGCTTCGGATCGGTAGTAGAAGA NB89 ACAGCATCAATGTTTGGCTAGTTG CAACTAGCCAAACATTGATGCTGT NB90 GATGTAGAGGGTACGGTTTGAGGC GCCTCAAACCGTACCCTCTACATC NB91 GGCTCCATAGGAACTCACGCTACT AGTAGCGTGAGTTCCTATGGAGCC NB92 TTGTGAGTGGAAAGATACAGGACC GGTCCTGTATCTTTCCACTCACAA NB93 AGTTTCCATCACTTCAGACTTGGG CCCAAGTCTGAAGTGATGGAAACT NB94 GATTGTCCTCAAACTGCCACCTAC GTAGGTGGCAGTTTGAGGACAATC NB95 CCTGTCTGGAAGAAGAATGGACTT AAGTCCATTCTTCTTCCAGACAGG NB96 CTGAACGGTCATAGAGTCCACCAT ATGGTGGACTCTATGACCGTTCAG
