- Materials
-
- 1 µg (or 100-200 fmol) high molecular weight genomic DNA
- OR 100+ ng high molecular weight genomic DNA if performing DNA fragmentation
- Ligation Sequencing Kit XL V14 (SQK-LSK114-XL)
- Consumables
-
- NEBNext® Companion Module for Oxford Nanopore Technologies® Ligation Sequencing (NEB, E7180S or E7180L).
Alternatively, you can use the NEBNext® products below:
- NEBNext FFPE Repair Mix (NEB, M6630)
- NEBNext Ultra II End repair/dA-tailing Module (NEB, E7546)
- NEBNext Quick Ligation Module (NEB, E6056)
- Agencourt AMPure XP beads (Beckman Coulter™ cat # A63881)
- Nuclease-free water (e.g. ThermoFisher, AM9937)
- Freshly prepared 80% ethanol in nuclease-free water
- Hamilton 50 µl CO-RE tips with filter (Cat# 235948)
- Hamilton 300 µl CO-RE tips with filter (Cat# 235903)
- Hamilton 1000 µl CO-RE tips with filter (Cat# 235905)
- Hamilton 60 ml Reagent Reservoir, Self-Standing with Lid (Cat# 56694-01)
- Hamilton PCR ComfortLid (Cat# 814300)
- Bio-Rad Hard-Shell® 96-Well PCR Plates (Cat# HSP9601)
- Hamilton 20 ml Reagent Reservoirs (Cat# 96424-02)
- Sarstedt Inc Screw Cap Micro tube 2 ml, PP 1000/case (e.g. FisherScientific, Cat# NC0418367)
- Thermo Scientific™ Abgene™ 96 Well 0.8 ml Polypropylene Deepwell Storage Plate (Thermo Scientific™, cat # AB0859)
- Equipment
-
- Ice bucket with ice
- Vortex mixer
- Microplate centrifuge, e.g. Fisherbrand™ Mini Plate Spinner Centrifuge (Fisher Scientific, 11766427)
- Hamilton NGS STAR 96 (NGS STAR with Multi-Probe Head 96)
- Hamilton On-Deck Thermal Cycler (ODTC)
- Optional equipment
-
- Agilent Bioanalyzer (or equivalent)
- Qubit fluorometer plate reader (or equivalent for QC check)
-
For this protocol, you will need 1 µg (100-200 fmol) of gDNA.
-
Input DNA
How to QC your input DNA
It is important to use a plate reader to ensure the input DNA meets the quantity and quality requirements. Using too little or too much DNA, or DNA of poor quality (e.g. highly fragmented or containing RNA or chemical contaminants) can affect your library preparation.
For instructions on how to perform quality control of your DNA sample, please read the Input DNA/RNA QC protocol.
-
Input file worklist
A worklist input Excel file is required prior to running the protocol on the Hamilton NGS STAR 96. These contain information regarding the appropriate number of samples and well identifiers.
Example:
Source_SampleIDSource_WellTarget_WellSample_01 A1 A1 Sample_02 B1 B2 Sample_03 C1 C1 -
Hamilton NGS STAR 96
This method has been tested and validated using the Hamilton NGS STAR 96 (with 8 channels and MPH96) including an on deck thermal cycler (ODTC). An option to not use the ODTC is available in the method. The protocol may require some fine tuning for the NGS STAR 96 setup and the temperature/humidity of the customer laboratory.
Please contact your Hamilton representative for further details.
Deck layout
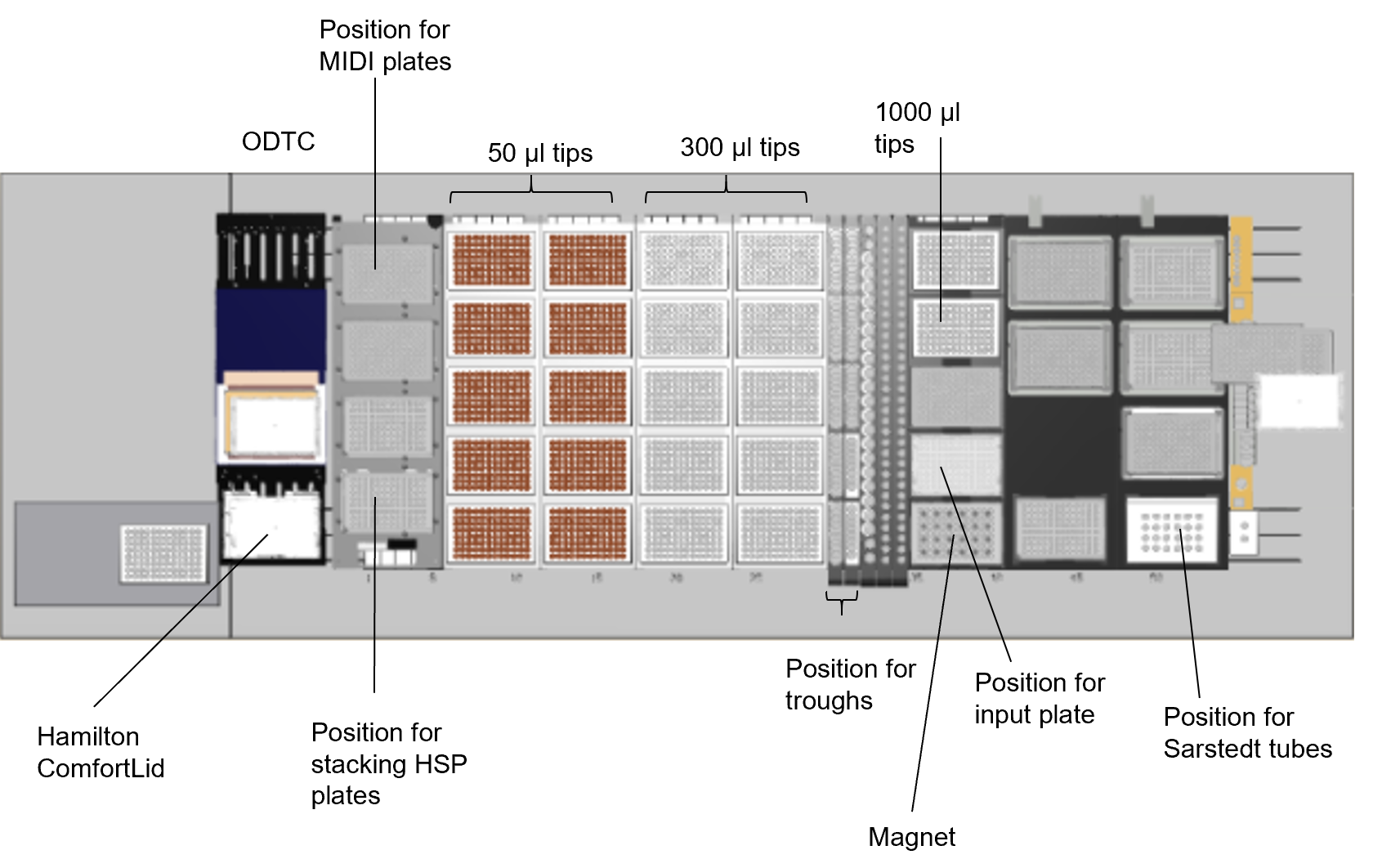
- ODTC: On-Deck Thermal Cycler Module
- MIDI plates: Abgene™ 96 Well 0.8mL Polypropylene Deepwell Storage Plate
- Troughs: Hamilton 20 ml Reagent Reservoirs
- HSP plates: Bio-Rad Hard-Shell® 96-Well PCR Plates
- Hamilton ComfortLid: Hamilton PCR ComfortLid
-
NEBNext® Companion Module for Oxford Nanopore Technologies® Ligation Sequencing
For customers new to nanopore sequencing, we recommend buying the NEBNext® Companion Module for Oxford Nanopore Technologies® Ligation Sequencing (catalogue number E7180S or E7180L), which contains all the NEB reagents needed for use with the Ligation Sequencing Kit.
Please note, for our amplicon protocols, NEBNext FFPE DNA Repair Mix and NEBNext FFPE DNA Repair Buffer are not required.
-
Third-party reagents
We have validated and recommend the use of all the third-party reagents used in this protocol. Alternatives have not been tested by Oxford Nanopore Technologies.
For all third-party reagents, we recommend following the manufacturer's instructions to prepare the reagents for use.
-
Consumables and reagent quantities required:
Consumables X24 samples X48 samples X96 samples Hamilton 50 µl CO-RE tips with filter 216 432 768 Hamilton 300 µl CO-RE tips with filter 451 694 1176 Hamilton 1000 µl CO-RE tips with filter 64 64 64 Hamilton 60 ml Reagent Reservoir, Self-Standing with Lid 3 3 3 Hamilton PCR ComfortLid 1 1 1 Bio-Rad Hard-Shell® 96-Well PCR Plate 2 2 2 Hamilton 20 ml Reagent Reservoirs 2 2 2 Sarstedt Inc Screw Cap Micro Tube 2ml 2 4 5 Abgene™ 96 Well 0.8mL Polypropylene Deepwell Storage Plate 2 2 2 Reagents/kits X24 samples X48 samples X96 samples 80% ethanol 16.5 ml 28 ml 51 ml AMPure XP Beads 6.8 ml 9.7 ml 15.5 ml Ligation Sequencing Kit XL V14 (SQK-LSK114-XL) 1 kit 1 kit 2 kits NEBNext Companion Module for Oxford Nanopore Technologies Ligation Sequencing (Cat# E7180L) 1 kits 1 kits 1 kits NEBNext Companion Module for Oxford Nanopore Technologies Ligation Sequencing (Cat# E7180S) - 1 kits 1 kits Alternatively: - - - NEBNext FFPE DNA Repair Mix (Cat# M6630L) 1 kit 1 kit 2 kits NEBNext Ultra II End Repair/dA-Tailing Module (Cat# E7546L) 1 kit 2 kits 3 kits NEBNext Quick Ligation Module (Cat# E6056L) 1 kit 2 kits 3 kits Note: These are the number of kits required for one run through for the selected number of samples.
-
Ligation Sequencing Kit XL V14 (SQK-LSK114-XL) contents
Name Acronym Vial colour Number of vials Fill volume per vial (µl) DNA Control Strand DCS Yellow 1 100 Ligation Adapter LA Green 1 320 Ligation Buffer LNB White 1 1,500 Elution Buffer EB White cap, black strip label 1 10,000 Long Fragment Buffer LFB White cap, orange strip label 2 20,000 Short Fragment Buffer SFB White cap, blue strip label 2 20,000 Library Beads LIB Pink 2 1,800 Library Solution LIS White cap, pink label 2 1,800 Sequencing Buffer SB Red 3 1,700 Flow Cell Flush FCF Clear 4 15,500 Flow Cell Tether FCT Purple 1 1,600 Note: The DNA Control Sample (DCS) is a 3.6 kb standard amplicon mapping the 3' end of the Lambda genome.
