- Materials
-
- Adapter Mix (AMX)
- Ligation Buffer (LNB)
- Long Fragment Buffer (LFB)
- Short Fragment Buffer (SFB)
- Elution Buffer (EB)
- Consumables
-
- Agencourt AMPure XP beads (Beckman Coulter™, A63881)
- NEBNext Quick Ligation Module (NEB, E6056)
- Thermo Scientific™ Abgene™ 96 Well 0.8 ml Polypropylene Deepwell Storage Plate (Thermo Scientific™, cat # AB0859)
- Sarstedt Inc Screw Cap Micro tube 2 ml, PP 1000/case (e.g. FisherScientific, Cat# NC0418367)
- Roche Diagnotics MagNA Pure LC Medium Reagent Tubs 20 (Cat# 03004058001)
- Bio-Rad Hard-Shell® 96-Well PCR Plates (Cat# HSP9601)
- Hamilton 60 ml Reagent Reservoir, Self-Standing with Lid (Cat# 56694-01)
- Hamilton 1000 µl CO-RE tips with filter (Cat# 235905)
- Hamilton 300 µl CO-RE tips with filter (Cat# 235903)
- Hamilton 50 µl CO-RE tips with filter (Cat# 235948)
- Equipment
-
- P1000 pipette and tips
- P200 pipette and tips
- P100 pipette and tips
- P10 pipette and tips
- Vortex mixer
- Microplate centrifuge, e.g. Fisherbrand™ Mini Plate Spinner Centrifuge (Fisher Scientific, 11766427)
-
Consumables and equipment quantities:
Consumable/equipment X24 samples X48 samples X96 samples Hamilton 50 µl CO-RE tips with filter 120 240 384 Hamilton 300 µl CO-RE tips with filter 250 396 686 Hamilton 1000 µl CO-RE tips with filter 32 32 32 Hamilton 60 ml Reagent Reservoir, Self-Standing with Lid 2 (1 L/SFB & 1 EB) 2 (1 L/SFB & 1 EB) 2 (1 L/SFB & 1 EB) Bio-Rad Hard-Shell® 96-Well PCR Plate 1 1 1 Hamilton 20 ml Reagent Reservoirs 1 1 1 Sarstedt Inc Screw Cap Micro Tube 2 ml 1 2 3 Abgene™ 96 Well 0.8 ml Polypropylene Deepwell Storage Plate 1 1 1 -
Reagents quantities:
Reagents X24 samples X48 samples X96 samples Adapter Mix (AMX) 2 tubes 4 tubes 8 tubes Ligation Buffer (LNB) 2 tubes 4 tubes 8 tubes Elution Buffer (EB) 1 bottle 1 bottle 1 bottle Long Fragment Buffer (LFB) 2 bottles 4 bottles 8 bottles Short Fragment Buffer (SFB) 2 bottles 4 bottles 8 bottles AMPure XP Beads 3.1 ml 4.3 ml 6.6 ml NEBNext Companion Module for Oxford Nanopore Technologies Ligation Sequencing (Cat# E7180S)
regarding the reagent below:2 tubes 3 tubes 5 tubes Alternatively: - - - Quick T4 DNA Ligase 1 tube 2 tubes 3 tubes Note: Dead volumes are included.
-
Spin down and store the Quick T4 Ligase on ice until use.
-
Spin down and combine all the required tubes of Adapter Mix (AMX) required, and place on ice.
-
Thaw the Ligation Buffer (LNB) at room temperature, spin down and combine all the required tubes. Place on ice immediately after thawing and mixing.
-
Thaw a bottle of Elution Buffer (EB) at room temperature, mix by vortexing and place on ice.
-
To enrich for DNA fragments of 3 kb or longer, thaw the Long Fragment Buffer (LFB) at room temperature, mix by vortexing and combine all the required bottles before storing on ice.
-
To retain DNA fragments of all sizes, thaw the Short Fragment Buffer (SFB) at room temperature, mix by vortexing and combine all the required bottles before storing on ice.
-
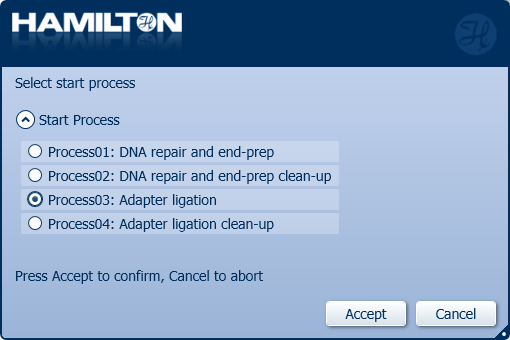
Click 'Process03: Adapter ligation' to start.
-
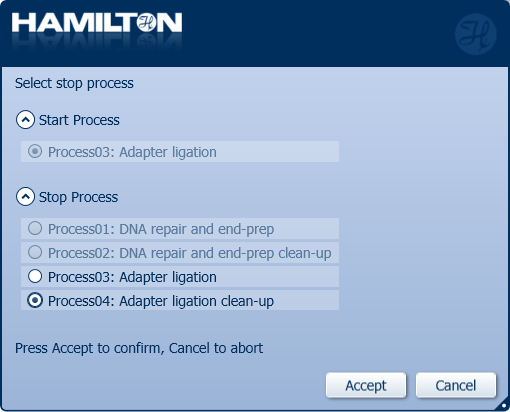
Click 'Process04: Adapter ligation and clean-up' to stop the automated library preparation and quantify the samples before sequencing.
-
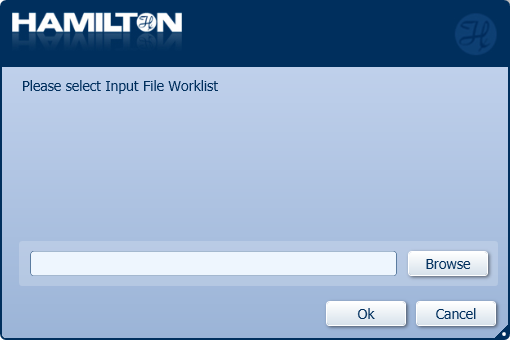
Click 'Browse' to choose the Input File Worklist used during DNA repair and end-prep.
-
Prepare the Adapter Ligation Mastermix with the following reagents according to the Hamilton user interface. Select either 'Yes' or 'No' to continue.
Note: It is user preference whether to print and save the instructions.
Reagent volumes for all sample numbers:
Reagent Volume X24 samples Volume X48 samples Volume X96 samples Adapter Mix (AMX) 140.5 µl 281 µl 559.5 µl Ligation Buffer (LNB) 702.5 µl 1405 µl 2797.5 µl Quick T4 DNA Ligase 281 µl 562 µl 1119 µl 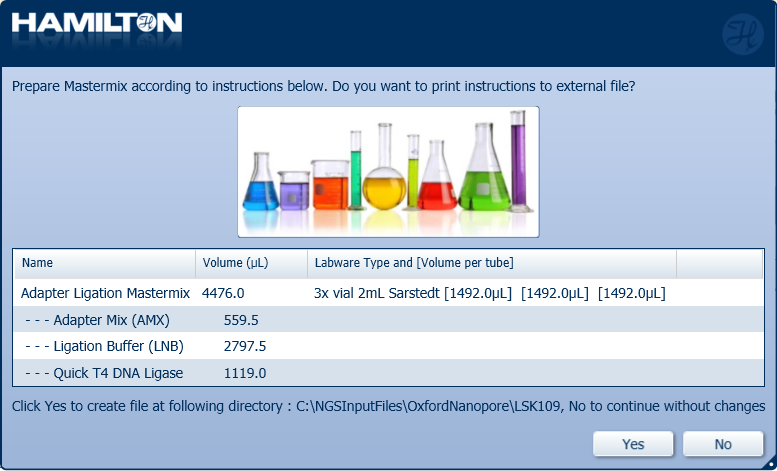
-
Insert plates to their corresponding positions on screen. Click 'Ok' to continue.
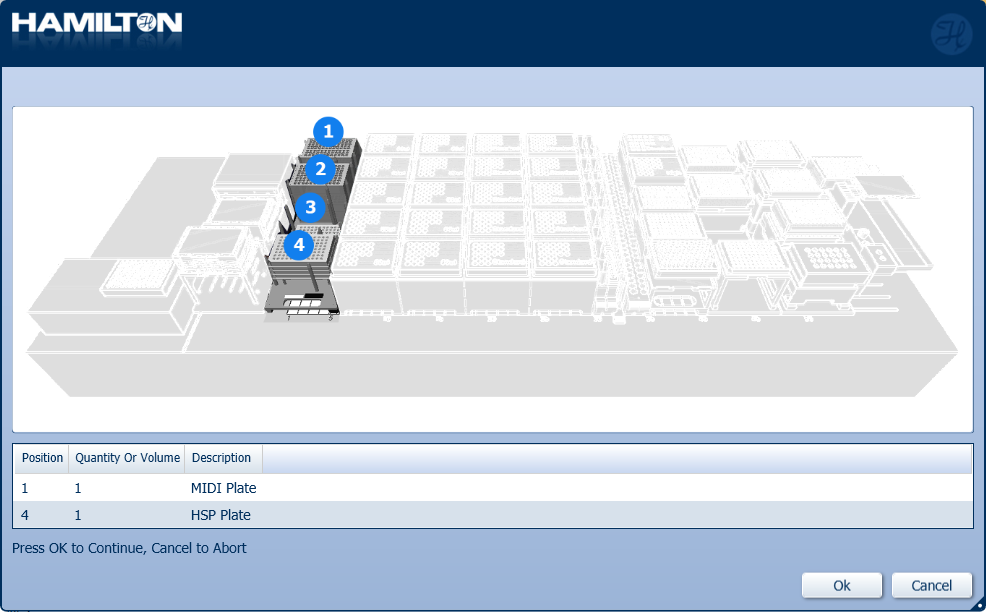
-
Load a full deck of 50 µl tips into the positions on screen. Click 'Ok' to continue.
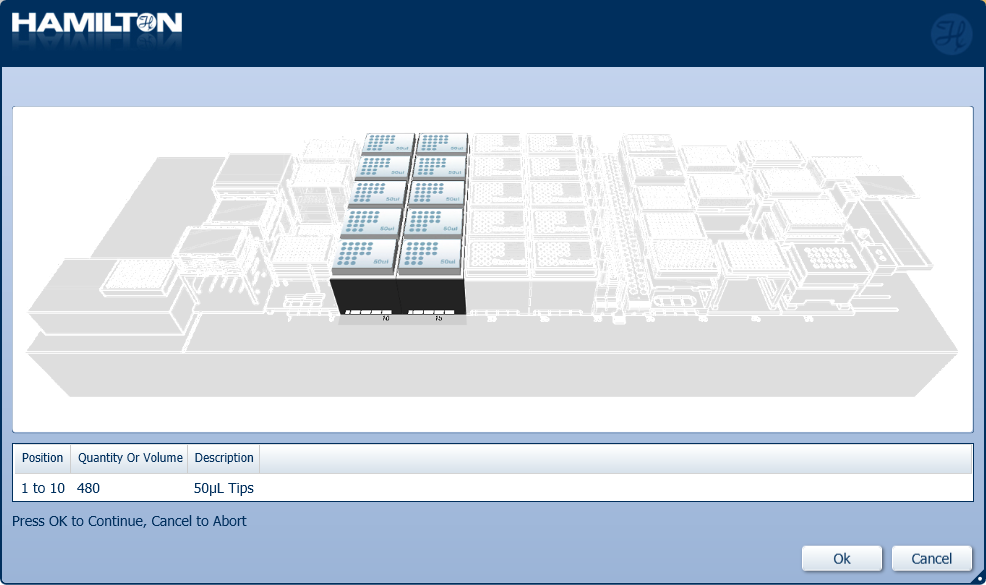
-
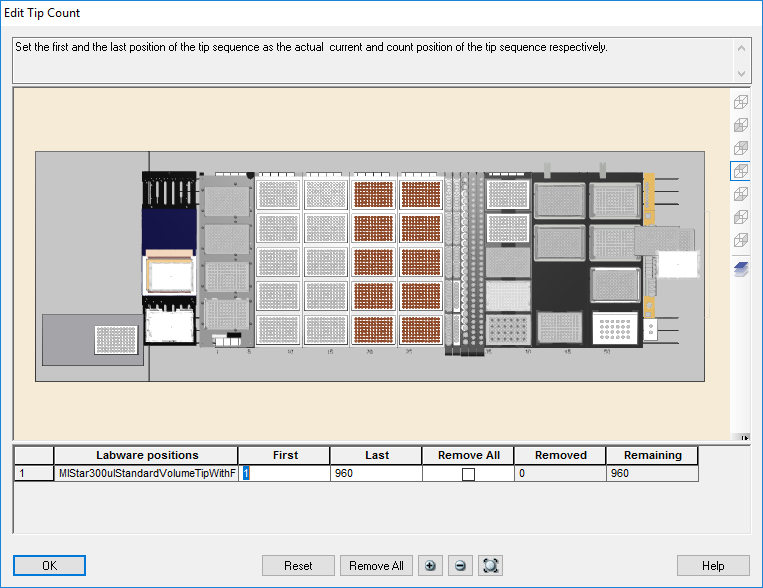
Highlight the 50 µl tips available to use on the 'Edit Tip Count' window. Click 'Ok' to continue.
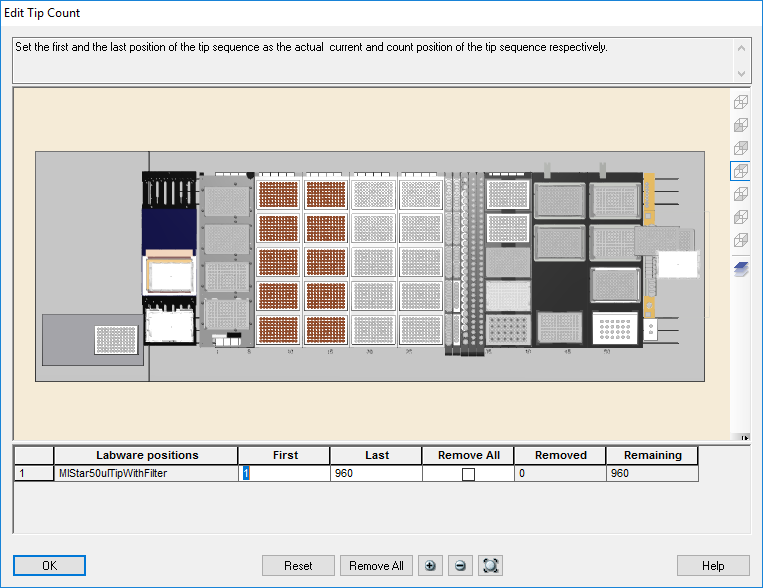
-
Load a full deck of 300 µl tips in the positions on screen. Click 'Ok' to continue.
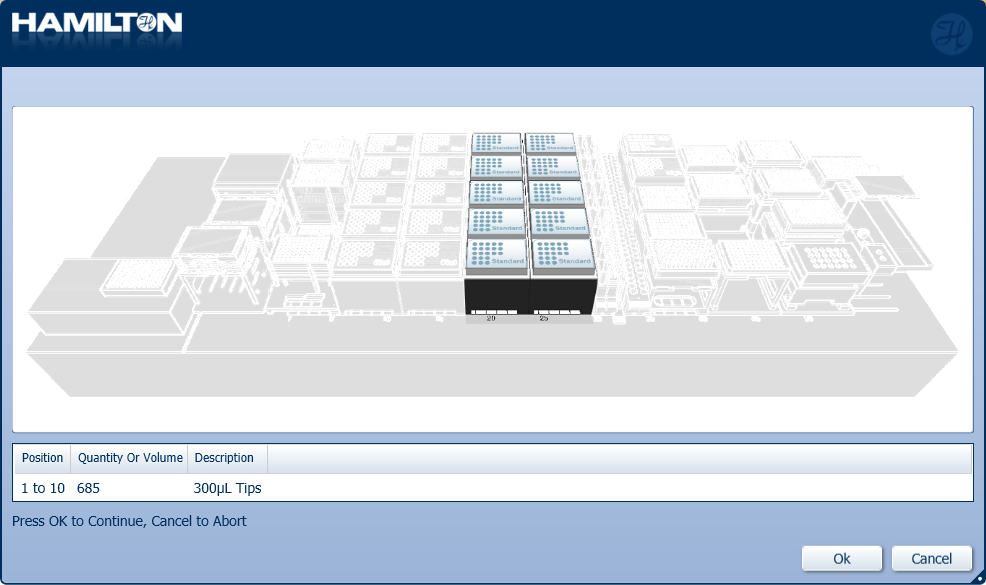
-
Highlight the 300 µl tips available to use on the 'Edit Tip Count' window. Click 'Ok' to continue.
-
Prepare the AMPure XP beads by vortexing and load the 20 ml trough with the volume required:
Reagents Volume X24 samples Volume X48 samples Volume X96 samples Beads 3.1 ml 4.3 ml 6.6 ml -
Insert troughs of AMPure XP beads, LFB/SFB and EB in the positions on screen. Click 'Ok' to continue.
Reagent Volume X24 samples Volume X48 samples Volume X96 samples Long/Short Fragment Buffer 2 bottles 4 bottles 8 bottles Elution Buffer 1 bottle 1 bottle 1 bottle 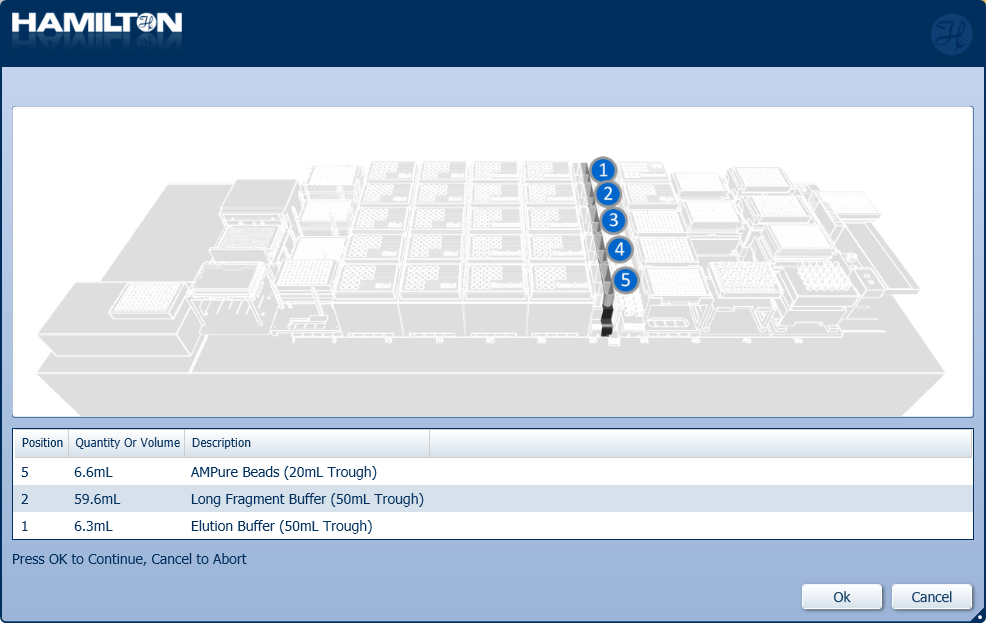
-
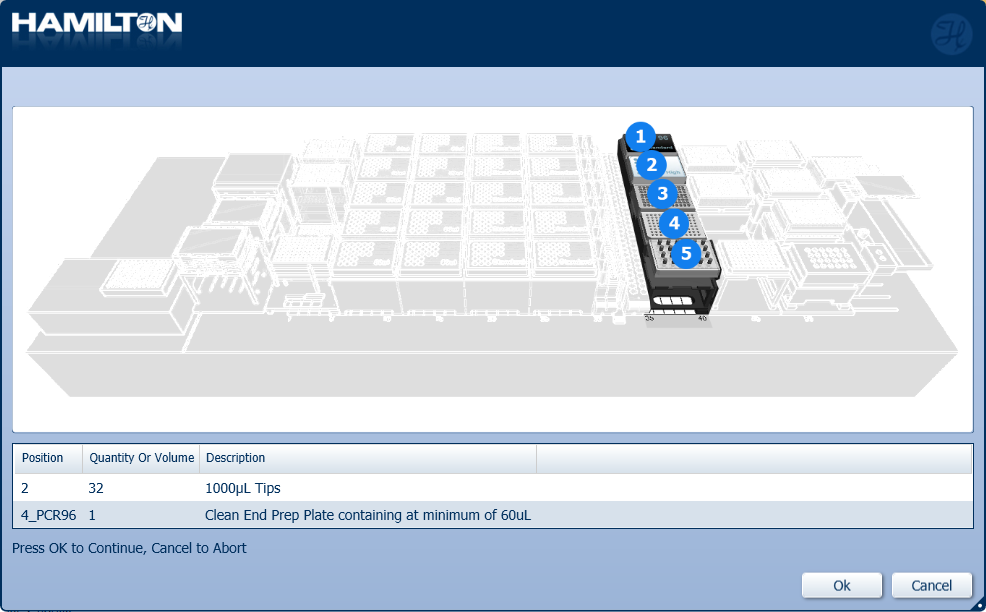
Insert 1000 µl tips and the Clean End Prep Plate to the correct positions on screen. Click 'Ok' to continue.
-
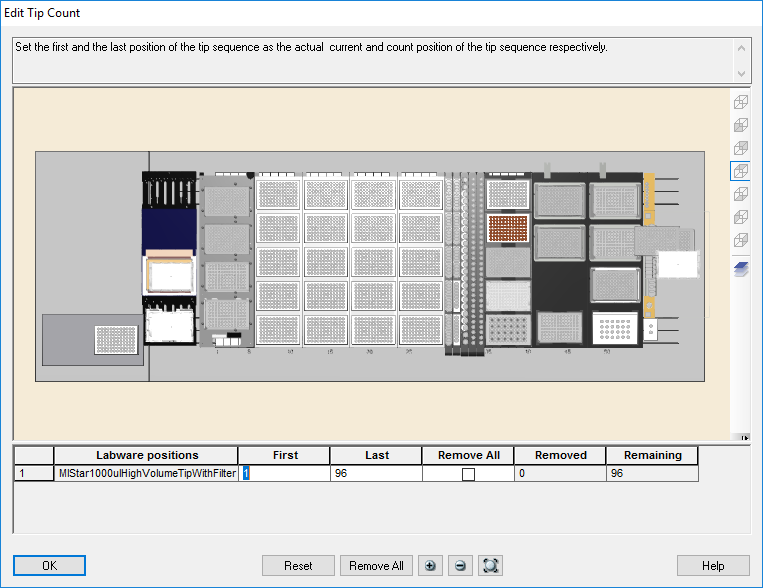
Highlight the 1000 µl tips available to use on the 'Edit Tip Count' window. Click 'Ok' to continue.
-
Insert the prepared Adapter Ligation Mastermix into the positions on screen. Click 'Ok' to continue.
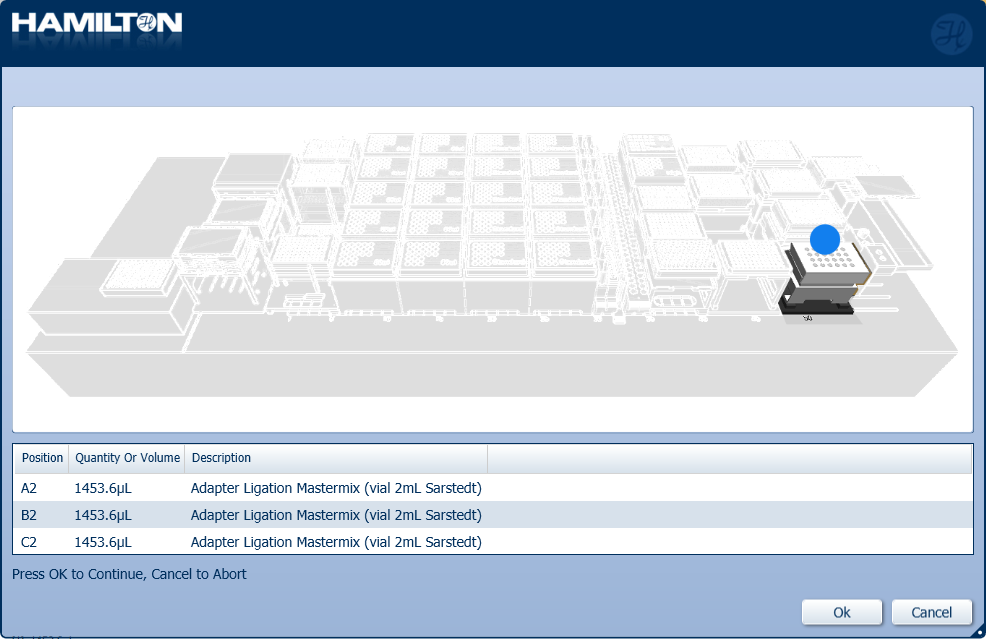
-
Once the automation process has finished, there will be an on screen prompt to unload the plate. Click 'Ok' to continue.

-
Quantify 1 µl of each eluted sample using a Qubit fluorometer plate reader off deck.
-
Optional actionIf quantities allow, the libraries may be diluted in Elution Buffer (EB) for splitting across multiple flow cells.
Additional buffer for doing this can be found in the Sequencing Auxiliary Vials expansion (EXP-AUX001), available to purchase separately. This expansion also contains additional vials of Sequencing Buffer (SQB) and Loading Beads (LB), required for loading the libraries onto flow cells.















