- Materials
-
- Flongle Sequencing Expansion (EXP-FSE002)
- Flow Cell Tether (FCT)
- Consumables
-
- Flongle Flow Cell
- 1.5 ml Eppendorf DNA LoBind tubes
- Equipment
-
- Flongle adapter
- MinION or GridION device
- P200 pipette and tips
- P20 pipette and tips
- P10 pipette and tips
-
The diagram below shows the components of the Flongle flow cell:
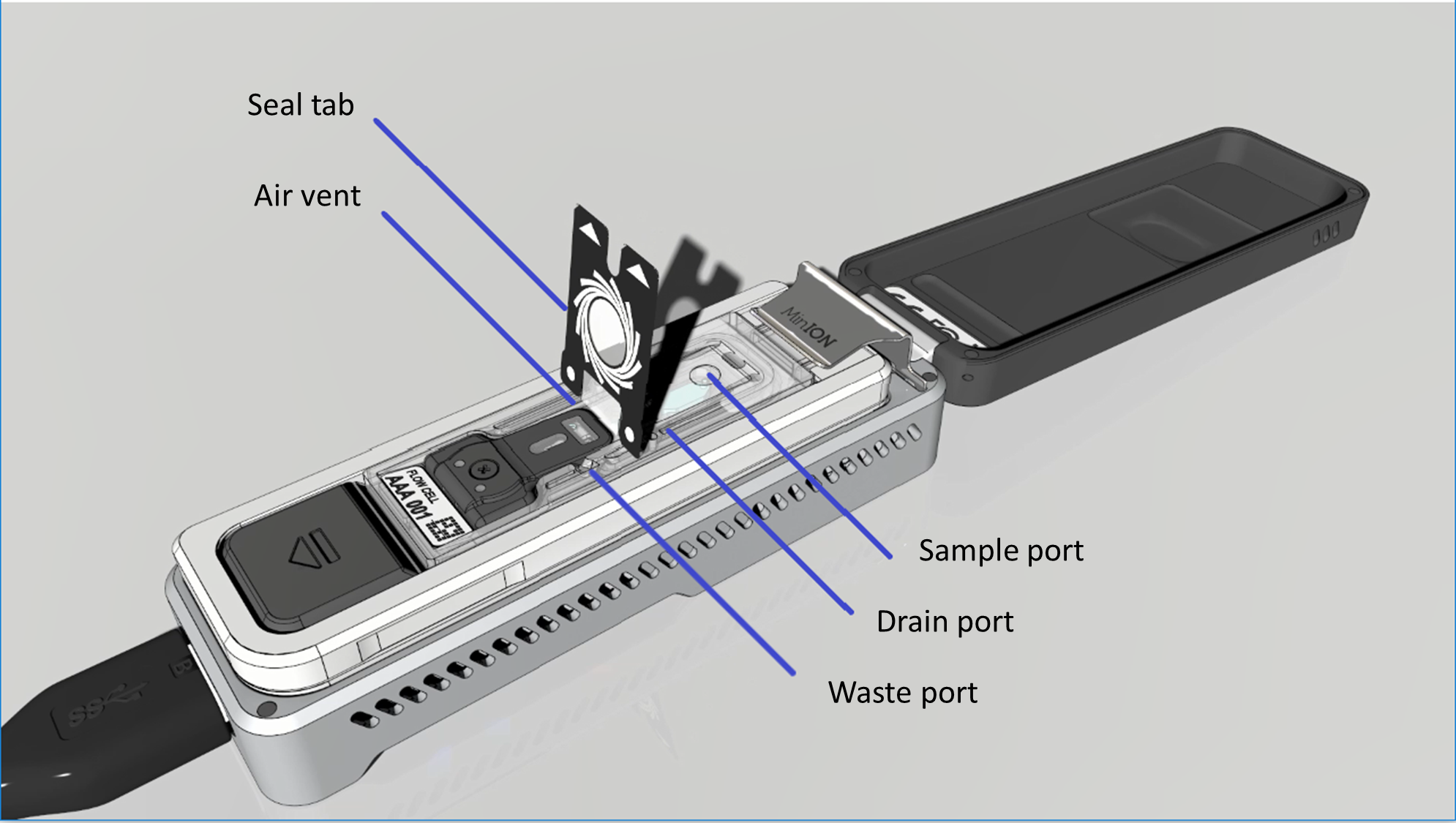
The seal tab, air vent, waste channel, drain port and sample port are visible here. The sample port, drain port and air vent only become accessible once the seal tab is peeled back.
-
Thaw the Sequencing Buffer (SB), Library Beads (LIB) or Library Solution (LIS, if using), Flow Cell Tether (FCT) and Flow Cell Flush (FCF) at room temperature before mixing by vortexing. Then spin down and store on ice.
-
In a fresh 1.5 ml Eppendorf DNA LoBind tube, mix 117 µl of Flow Cell Flush (FCF) with 3 µl of Flow Cell Tether (FCT) and mix by pipetting.
-
Place the Flongle adapter into the MinION or one of the five GridION positions.
The adapter should sit evenly and flat on the MinION Mk1B or GridION platform. This ensures the flow cell assembly is flat during the next stage.
-
Place the flow cell into the Flongle adapter, and press the flow cell down until you hear a click.
The flow cell should sit evenly and flat inside the adapter, to avoid any bubbles forming inside the fluidic compartments.

-
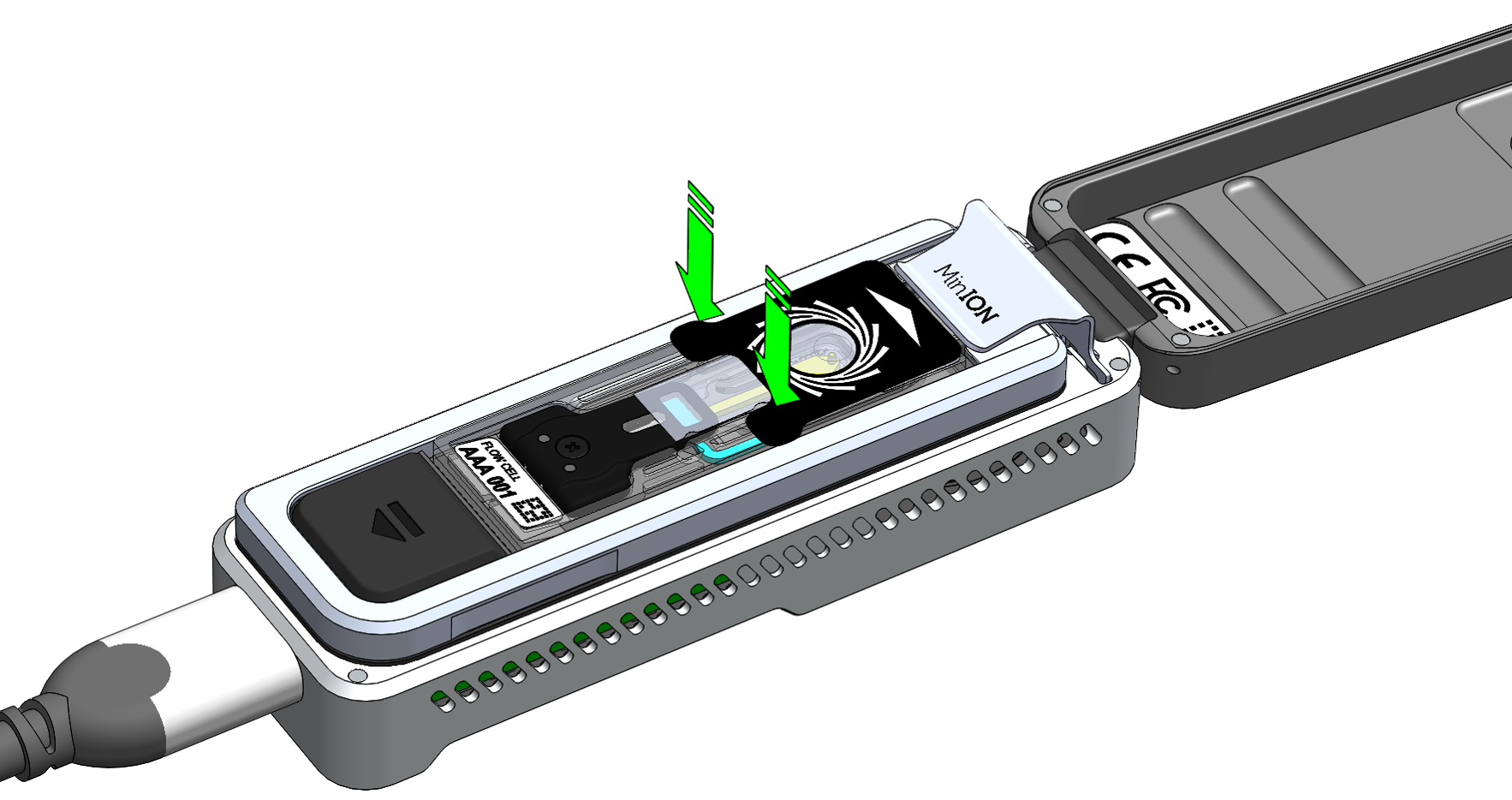
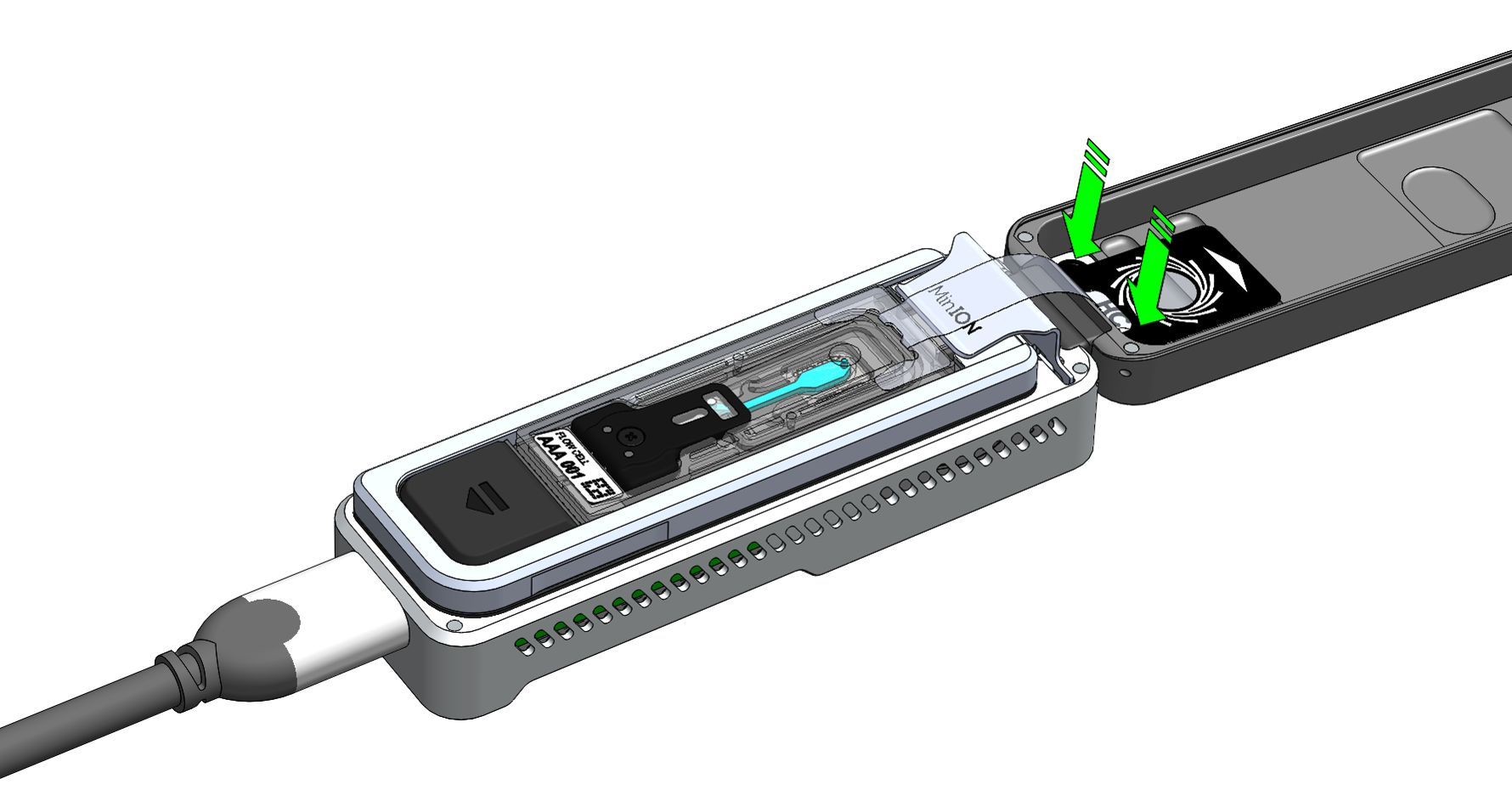
Peel back the seal tab from the Flongle flow cell, up to a point where the sample port is exposed, as follows:
Lift up the seal tab:
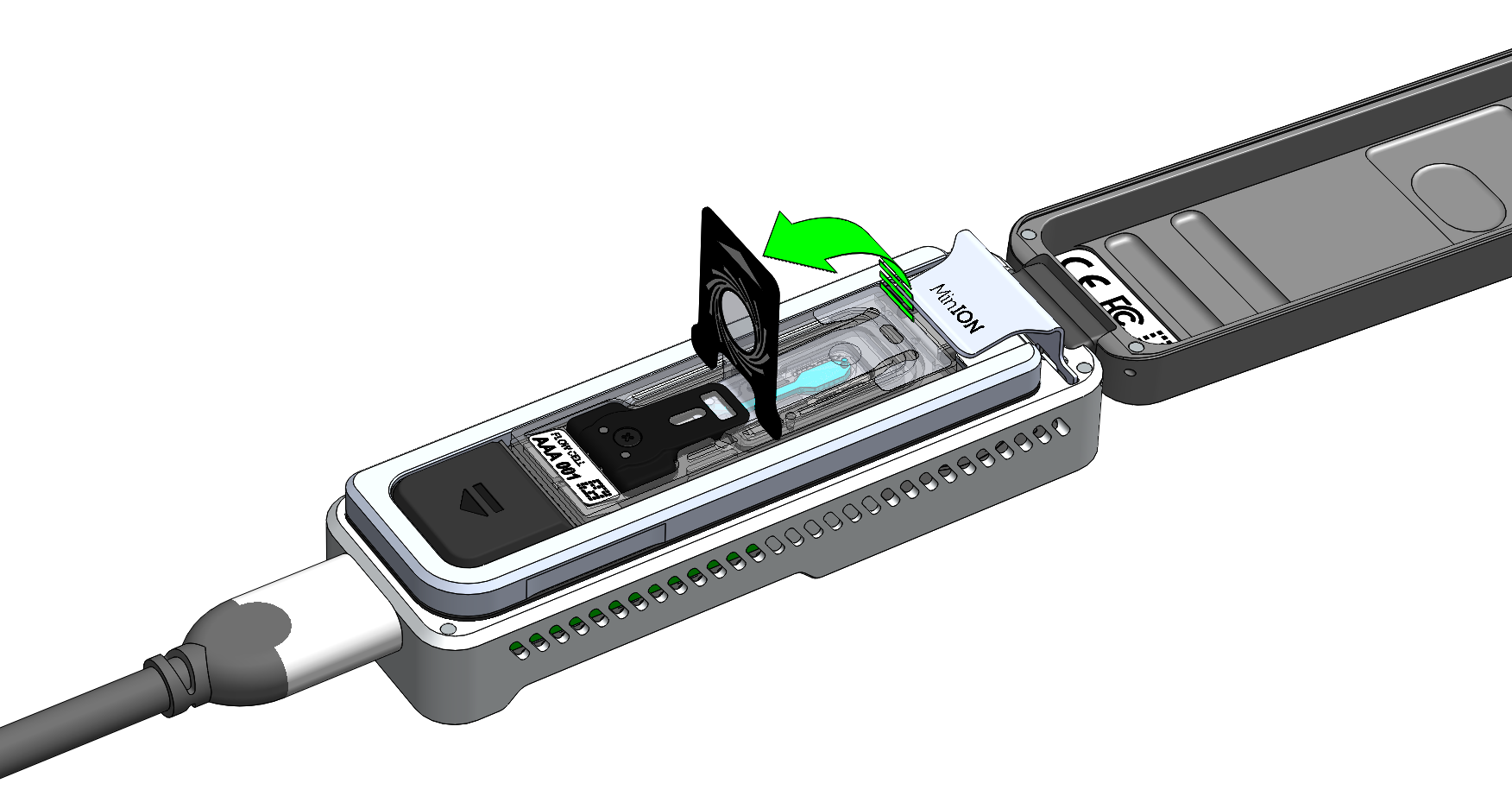
Pull the seal tab to open access to the sample port:

Hold the seal tab open by using adhesive on the tab to stick to the MinION Mk 1B lid:
-
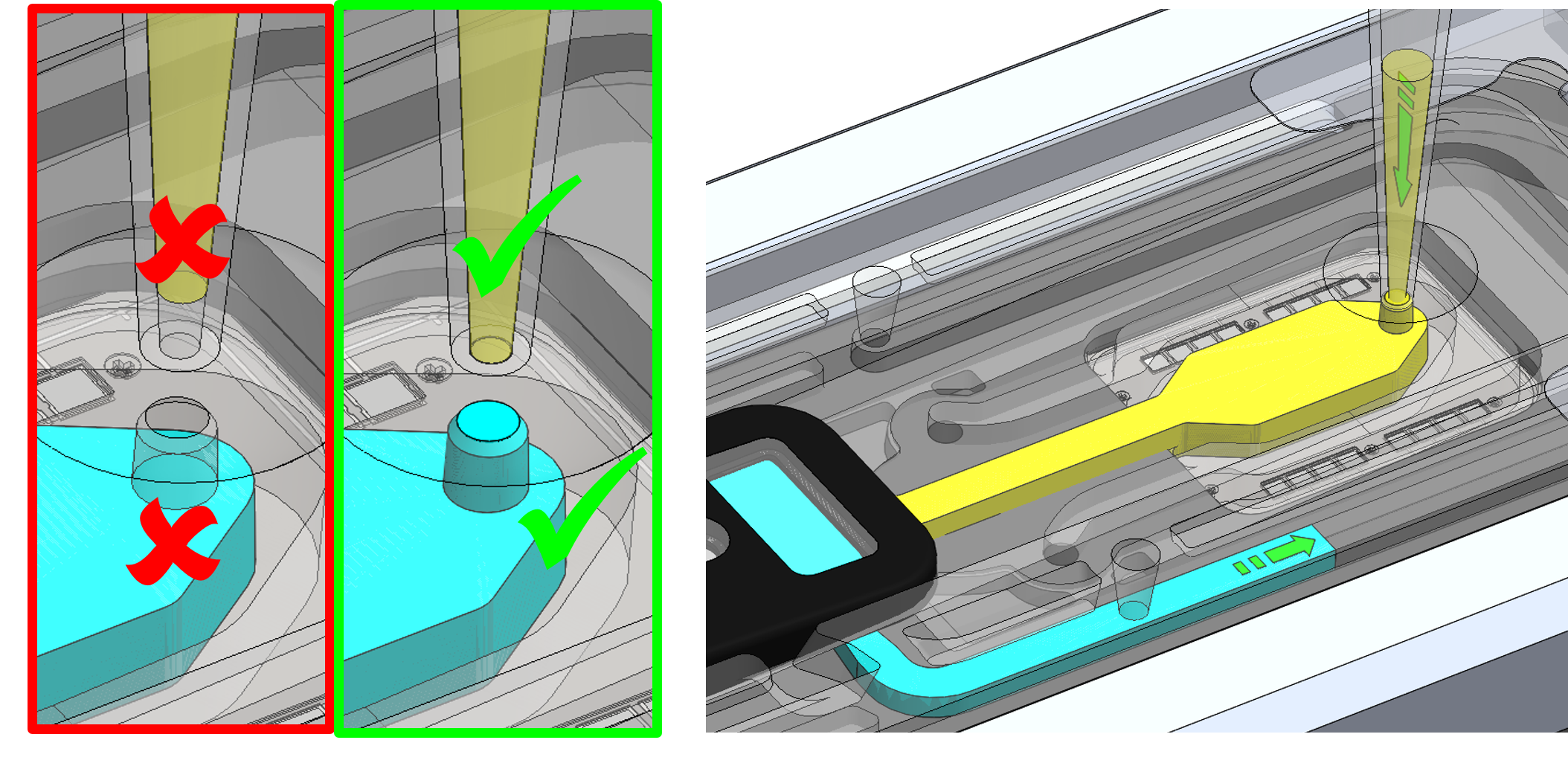
To prime your flow cell with the mix of Flow Cell Flush (FCF) and Flow Cell Tether (FCT) that was prepared earlier, ensure that there is no air gap in the sample port or the pipette tip. Place the P200 pipette tip inside the sample port and slowly dispense the 120 µl of priming fluid into the Flongle flow cell by slowly pipetting down. We also recommend twisting the pipette plunger down to avoid flushing the flow cell too vigorously.
-
Vortex the vial of Library Beads (LIB). Note that the beads settle quickly, so immediately prepare the Sequencing Mix in a fresh 1.5 ml Eppendorf DNA LoBind tube for loading the Flongle, as follows:
Reagents Volume Sequencing Buffer (SB) 15 µl Library Beads (LIB) mixed immediately before use, or Library Solution (LIS), if using. 10 µl DNA library 5 µl Total 30 µl -
To add the Sequencing Mix to the flow cell, ensure that there is no air gap in the sample port or the pipette tip. Place the P200 tip inside the sample port and slowly dispense the Sequencing Mix into the flow cell by slowly pipetting down. We also recommend twisting the pipette plunger down to avoid flushing the flow cell too vigorously.

-
Seal the Flongle flow cell using the adhesive on the seal tab, as follows:
Stick the transparent adhesive tape to the sample port.
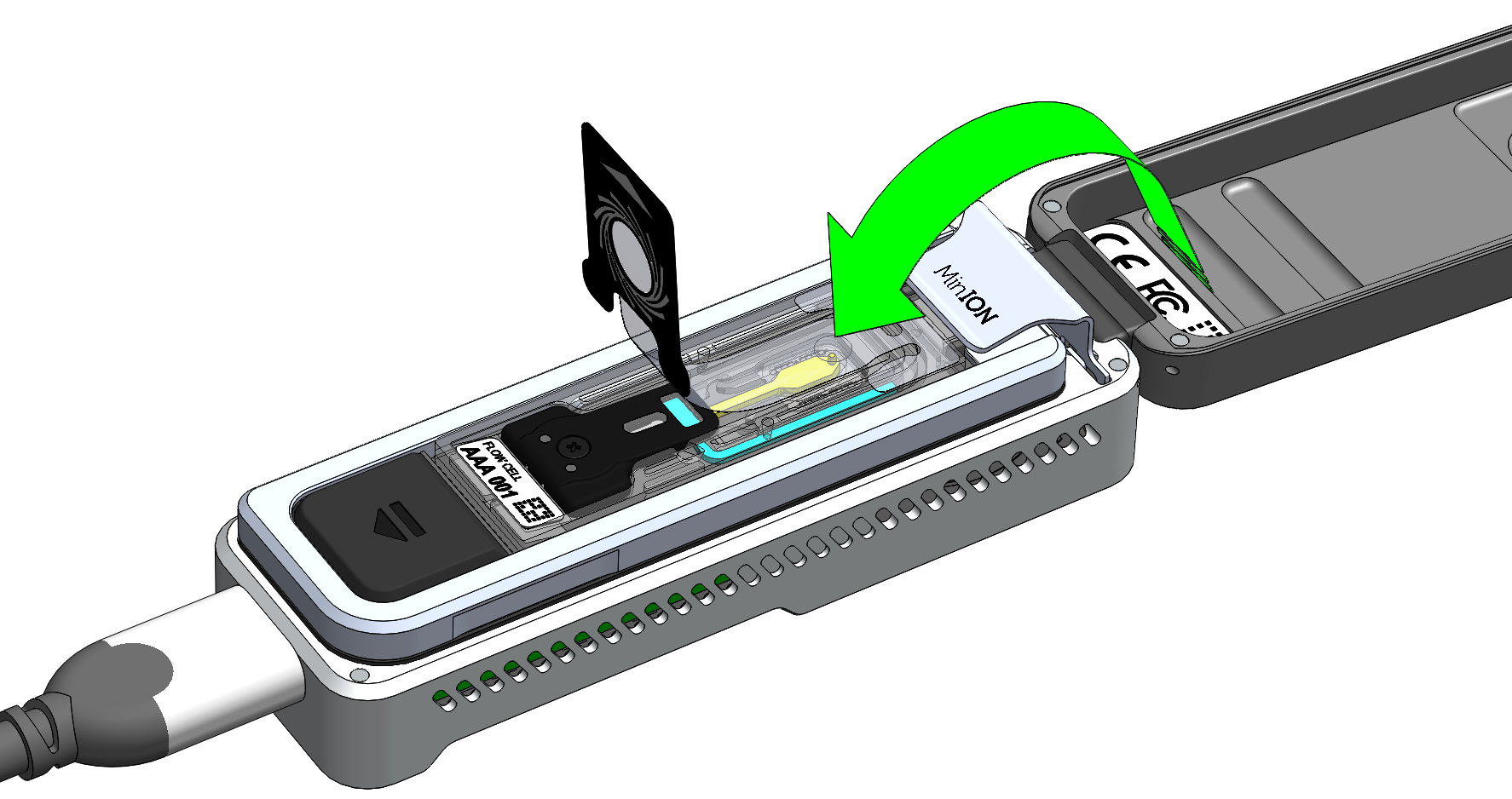
Replace the top (Wheel icon section) of the seal tab to its original position.