- Materials
-
- Input RNA in 10 mM Tris-HCl, pH 8.0
- LunaScript RT SuperMix (LS RT)
- Midnight Primer Pool A (MP A)
- Midnight Primer Pool B (MP B)
- Rapid Barcode Plate (RB96)
- AMPure XP Beads (AXP, or SPRI)
- Consumables
-
- Q5 HS Master Mix (Q5)
- Hard-Shell® 96-Well PCR Plates, low profile, thin-walled, skirted, white/clear (Bio-Rad, Cat # HSP9601)
- 96-well 0.8 ml MIDI plate (we recommend Abgene™ 96 Well 0.8 ml Polypropylene Deepwell Storage Plate: ThermoFisher, Cat # AB0859)
- Hamilton 50 µl CO-RE tips with filter (Cat# 235948)
- Hamilton 300 µl CO-RE tips with filter (Cat# 235903)
- Hamilton 1000 µl CO-RE tips with filter (Cat# 235905)
- Hamilton 60 ml Reagent Reservoir, Self-Standing with Lid (Cat# 56694-01)
- Hamilton PCR ComfortLid (Cat# 814300)
- 1.5 ml Eppendorf DNA LoBind tubes
- Equipment
-
- Hamilton NGS STAR liquid handling robot
- Hamilton On-Deck Thermal Cycler (ODTC)
- P1000 pipette and tips
- P200 pipette and tips
- P20 pipette and tips
- P2 pipette and tips
-
Consumables and equipment quantities:
Consumable/equipment X24 samples X48 samples X96 samples Hamilton 50 µl CO-RE tips with filter 274 491 925 Hamilton 3000 µl CO-RE tips with filter 30 60 120 Hamilton 1000 µl CO-RE tips with filter 3 6 12 Hamilton PCR ComfortLid 1 1 1 Hamilton 60 ml Reagent Reservoir, Self-Standing with Lid 2 2 2 MIDI plate 1 1 1 Bio-Rad Hard-Shell® 96-Well PCR Plate 2 2 3 -
Reagents quantities:
Reagent Volume X24 samples Volume X48 samples Volume X96 samples LunaScript RT SuperMix 62.8 µl 123.6 µl 251.2 µl Q5 Hot-start Master Mix 350 µl 700 µl 1400 µl Midnight Primer Pool A (MP A) 1.6 µl 3.2 µl 6.4 µl Midnight Primer Pool B (MP B) 1.6 µl 3.2 µl 6.4 µl Nuclease-free water 3,235 µl 3,570 µl 4,191 µl Elution Buffer 45.5 µl 57.6 µl 57.7 µl Rap F 4.2 µl 5.3 µl 5.3 µl SPRI beads 294 µl 588 µl 1176 µl 80% ethanol 8,400 µl 8,400 µl 8,400 µl -
Switch on the Hamilton NGS STAR 96 robot and open 'Hamilton Run Control' on the computer by clicking the icon:
-
Click 'File' and 'Open' to choose the method to run on the liquid handling robot.
-
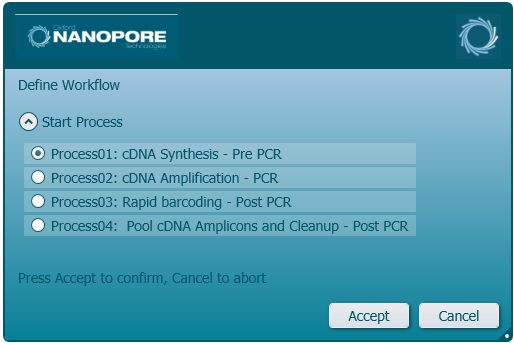
Click 'Process01: cDNA Synthesis - Pre-PCR' for the first process selection.
-
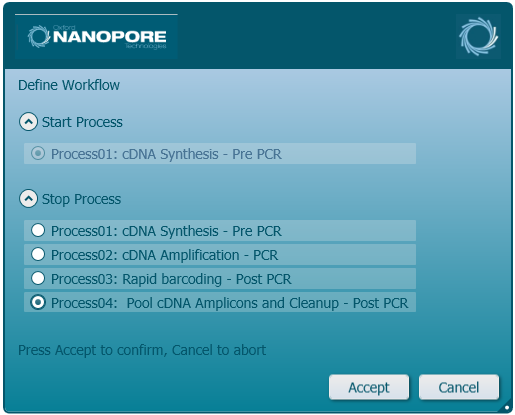
Click 'Process04: Pool cDNA Amplicons and Cleanup - Post PCR' as the last process selection.
-
Click 'Browse' to choose the Input File Worklist for the specific number of samples in the run and click 'OK'.
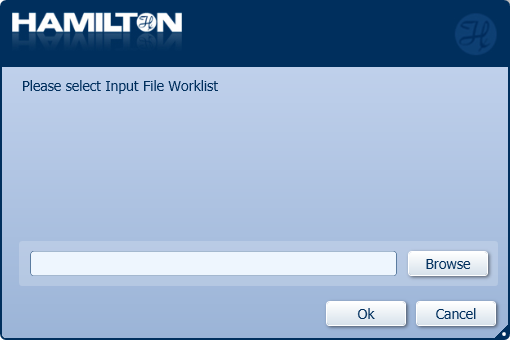
This will be a file provided with the script that passes information to the method including sample number and which wells of the rapid barcode plate to use during the rapid barcoding step.
-
Select 'Yes' for the on-deck thermal cycler (ODTC) use:
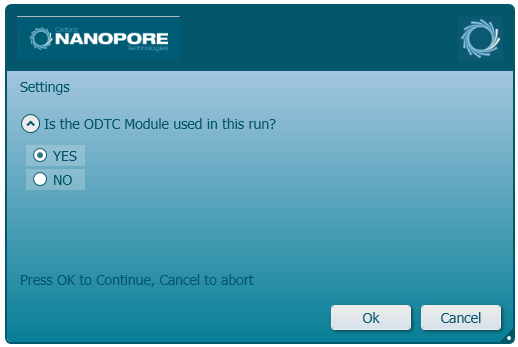
If selecting 'No', all steps using a thermal cycler will need to be performed manually off-deck.
-
Specify what well of the MIDI plate the final elution pool will result in by entering a number into the dialog:
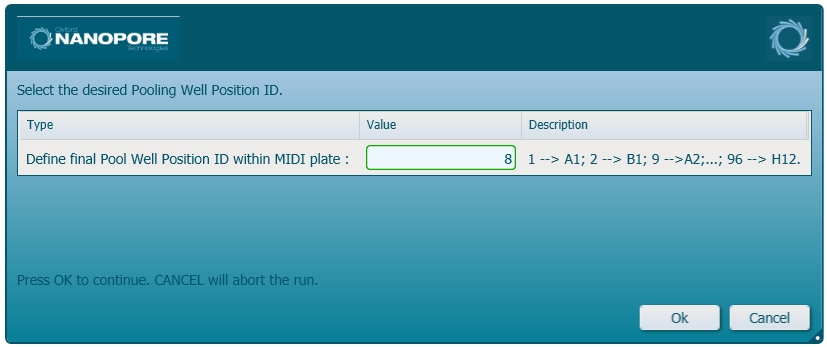
Important: The number introduced will correspond to the well of the plate when ordered by columns:

-
In a template-free pre-PCR hood, prepare the reagents according to the Hamilton user interface in 1.5 ml Eppendorf tubes and mix thoroughly. Click 'Ok' to continue.
Note: It is user preference whether to print and save the instructions.
The Midnight Primer Pool A (MP A) and Midnight Primer Pool B (MP B) are diluted in nuclease-free water prior to loading into the Hamilton deck.
Similarly, the Elution Buffer and Rap F will be combined prior to loading.Midnight primer mix preparation
For x24 samples:
Reagent Midnight Primer Mix 1 Midnight Primer Mix 1 Nuclease-free water 117.5 µl 117.5 µl Midnight Primer Pool A (MP A) 1.6 µl - Midnight Primer Pool B (MP B) - 1.6 µl Total 119.1 µl 119.1 µl For x48 samples:
Reagent Midnight Primer Mix 1 Midnight Primer Mix 1 Nuclease-free water 234.9 µl 234.9 µl Midnight Primer Pool A (MP A) 3.2 µl - Midnight Primer Pool B (MP B) - 3.2 µl Total 238.1 µl 238.1 µl For x96 samples:
Reagent Midnight Primer Mix 1 Midnight Primer Mix 1 Nuclease-free water 469.7 µl 469.7 µl Midnight Primer Pool A (MP A) 6.4 µl - Midnight Primer Pool B (MP B) - 6.4 µl Total 476.1 µl 476.1 µl EBRAP master mix preparation
Reagent Volume X24 samples Volume X48 samples Volume X96 samples Elution Buffer 45.5 µl 57.6 µl 57.7 µl Rap F 4.2 µl 5.3 µl 5.3 µl Total 49.7 µl 62.9 µl 63 µl Hamilton display:
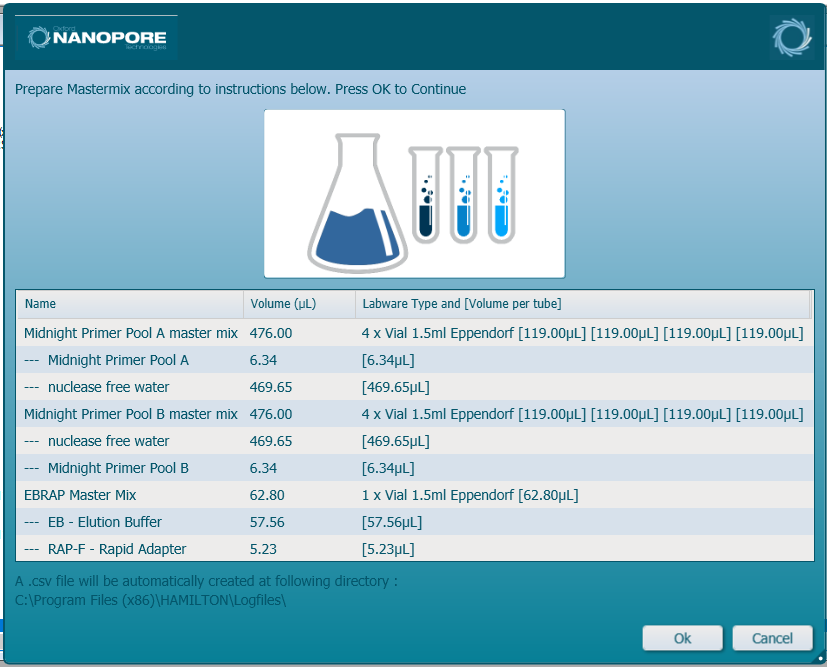
-
Insert the ComfortLid position as displayed on screen. Click 'Ok' to continue.
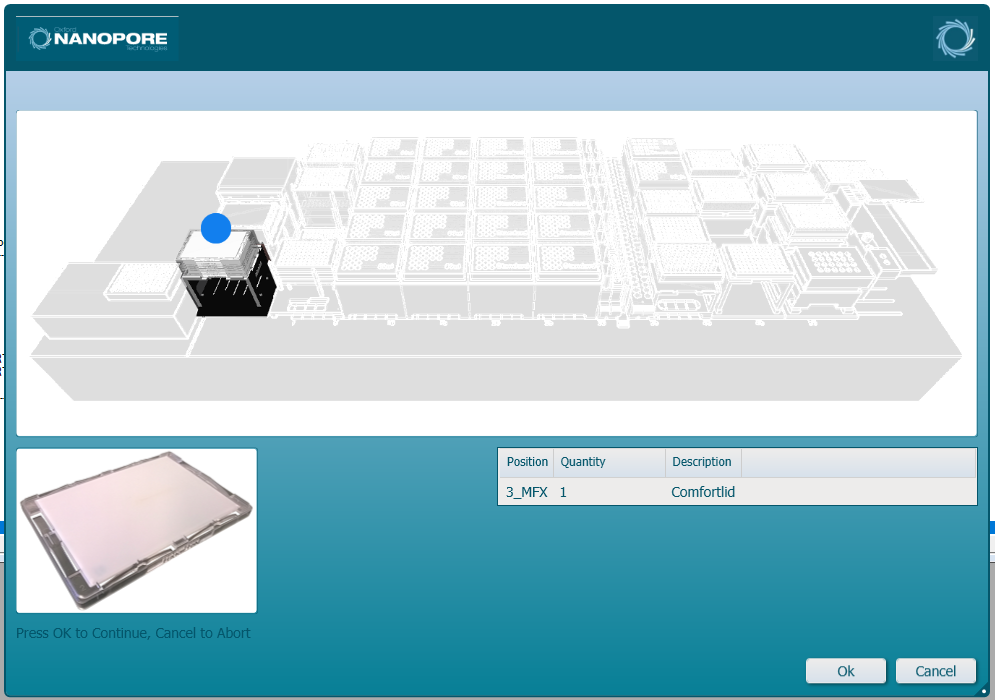
-
Load the microplates and the MIDI plates into the corresponding positions. Click 'Ok' to continue.
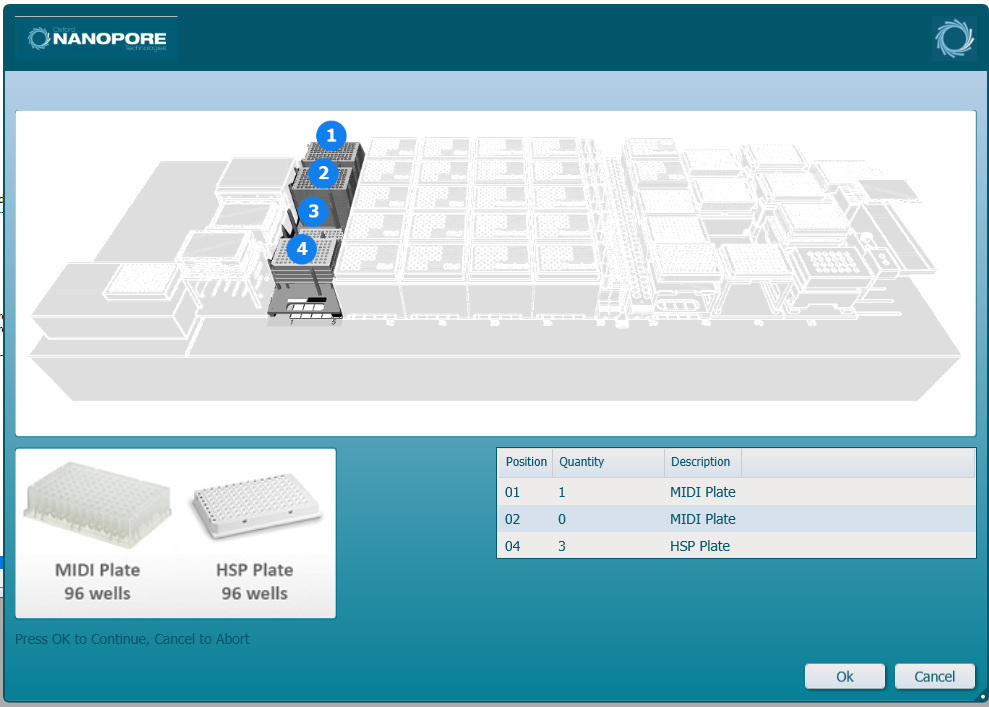
-
Acknowledge the pre-tip loading message to ensure you load all of the tips required the your run. Click 'Ok' to continue.

-
Load the LVF 50 µl tips into the positions on screen. Click 'Ok' to continue.
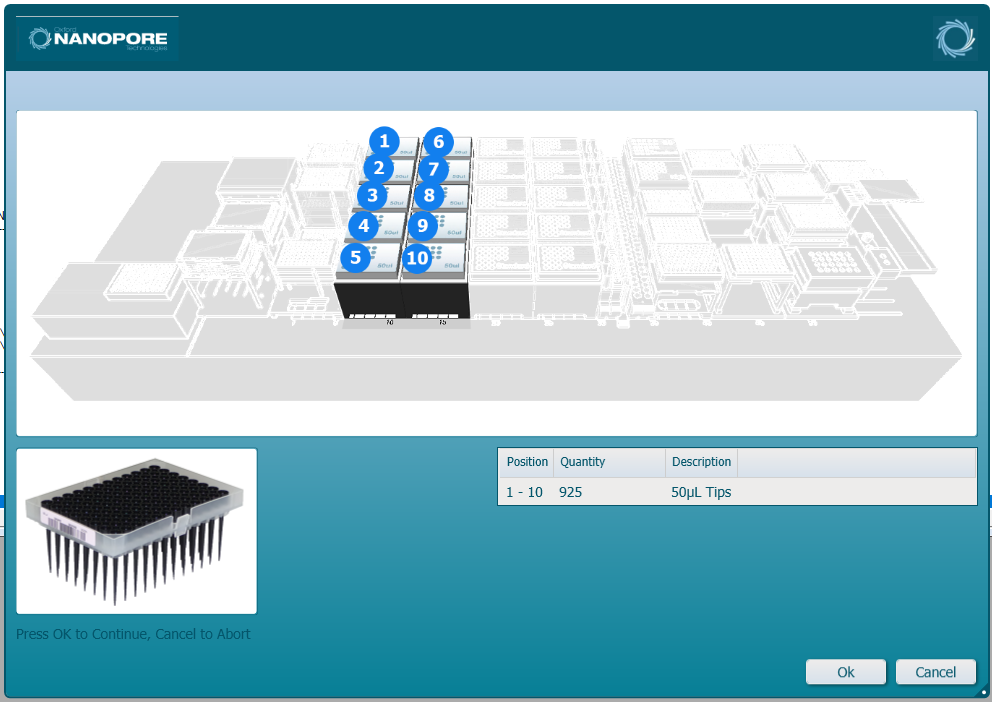
-
Highlight the 50 µl tips available to use on the 'Edit Tip Count' window and click 'Ok' to continue.
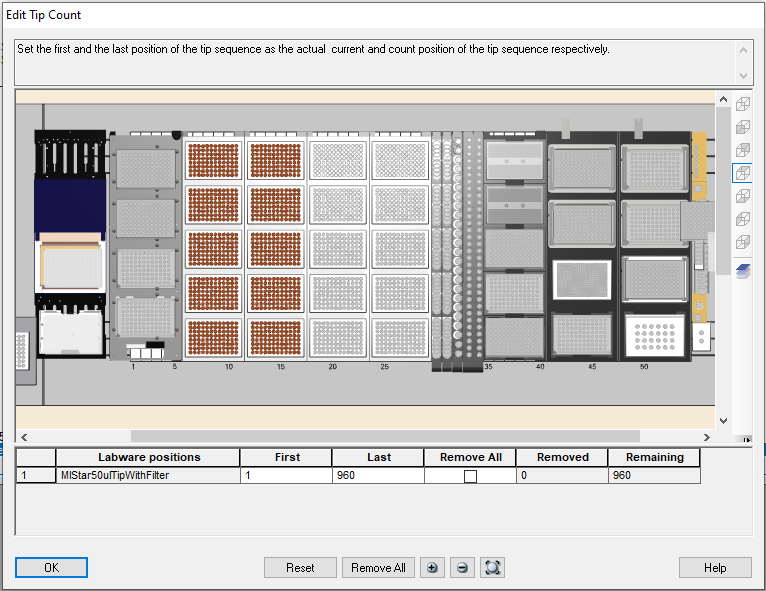
-
Load the SVF 300 µl tips into the positions on screen. Click 'Ok' to continue.
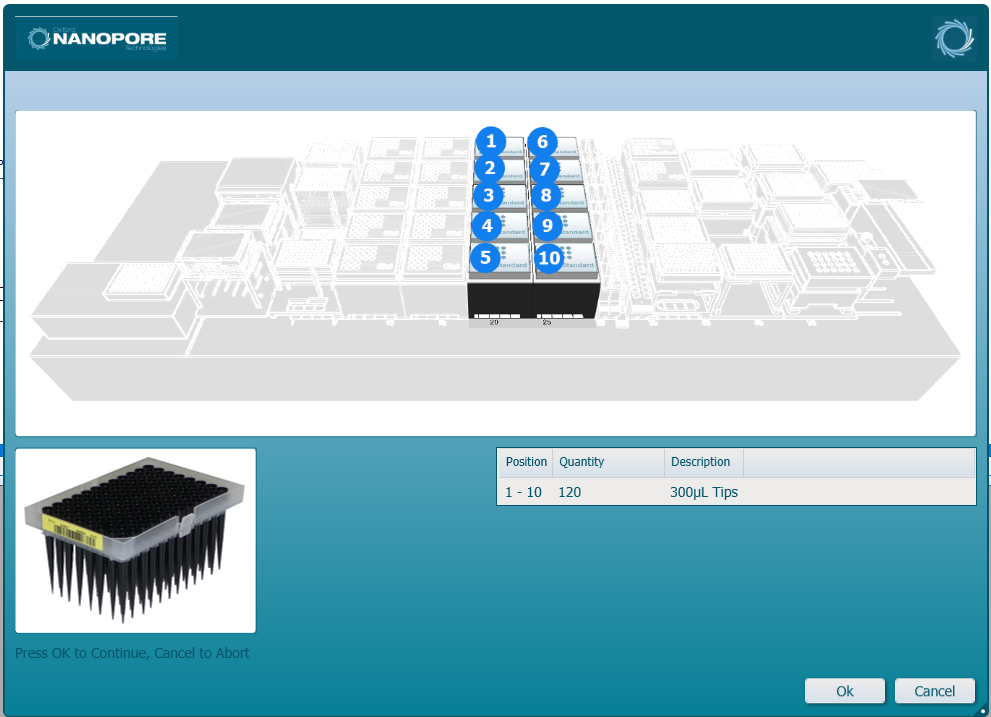
-
Highlight the 300 µl tips available to use on the 'Edit Tip Count' window and click 'Ok' to continue.
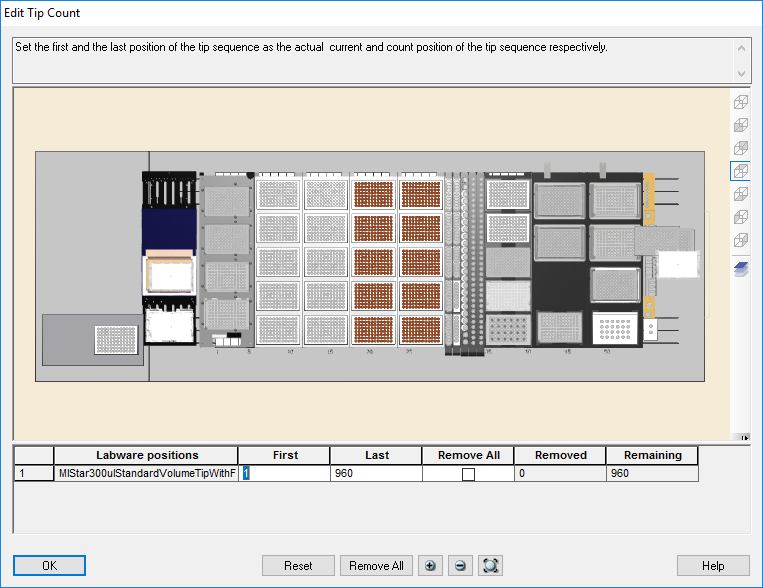
-
Freshly prepare 8.4 ml of 80% ethanol in nuclease-free water in a trough.
-
Load the reagent containers with the indicated fill volume for each trough.
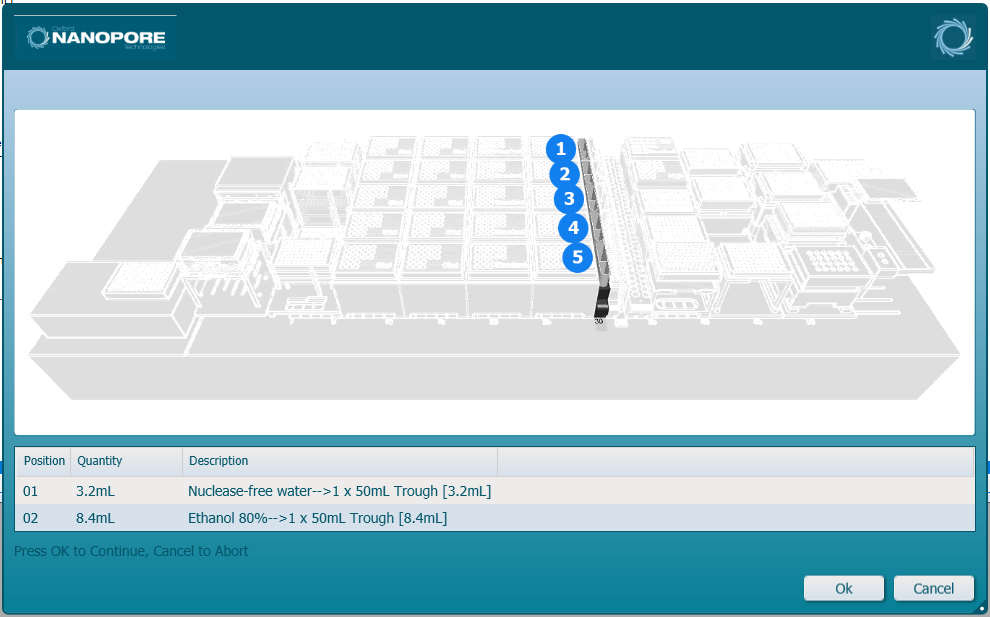
-
Load the HFV 1000 µl tips and insert the input plate of samples into the position on screen. Click 'Ok' to continue.
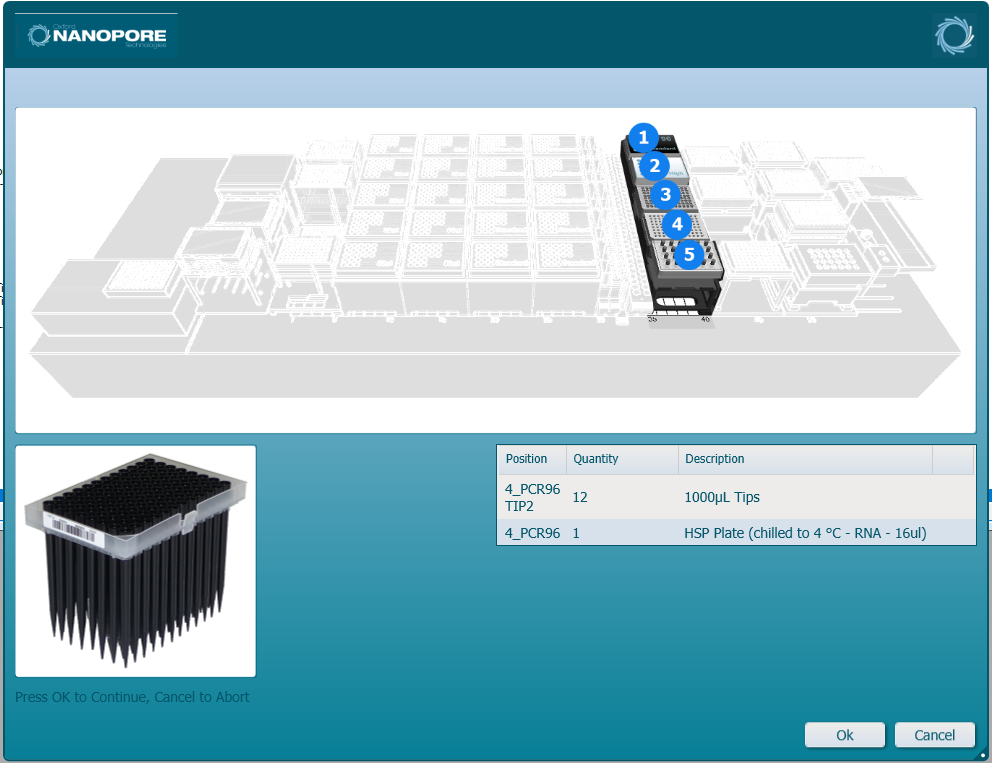
-
Highlight the 1000 µl tips available to use on the 'Edit Tip Count' window and click 'Ok' to continue.
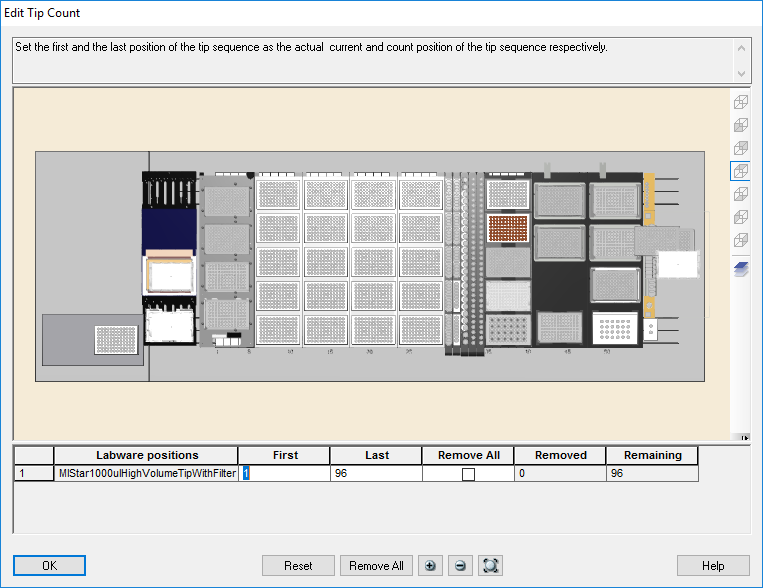
-
Load the CPAC chilled position and following the prompts on screen, load the required volume of reagents into their designated tube positions.
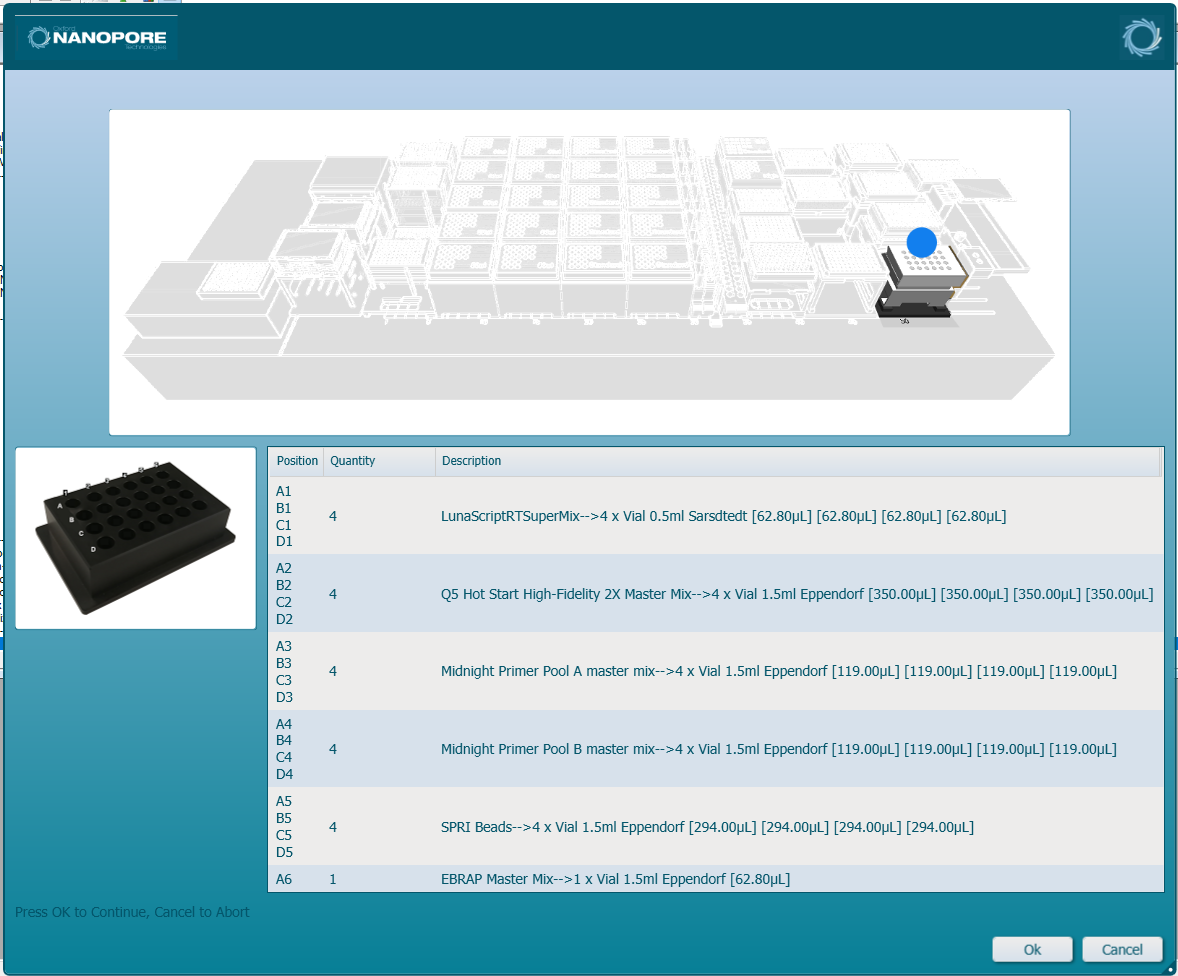
Tube layout in CPAC chilled position:
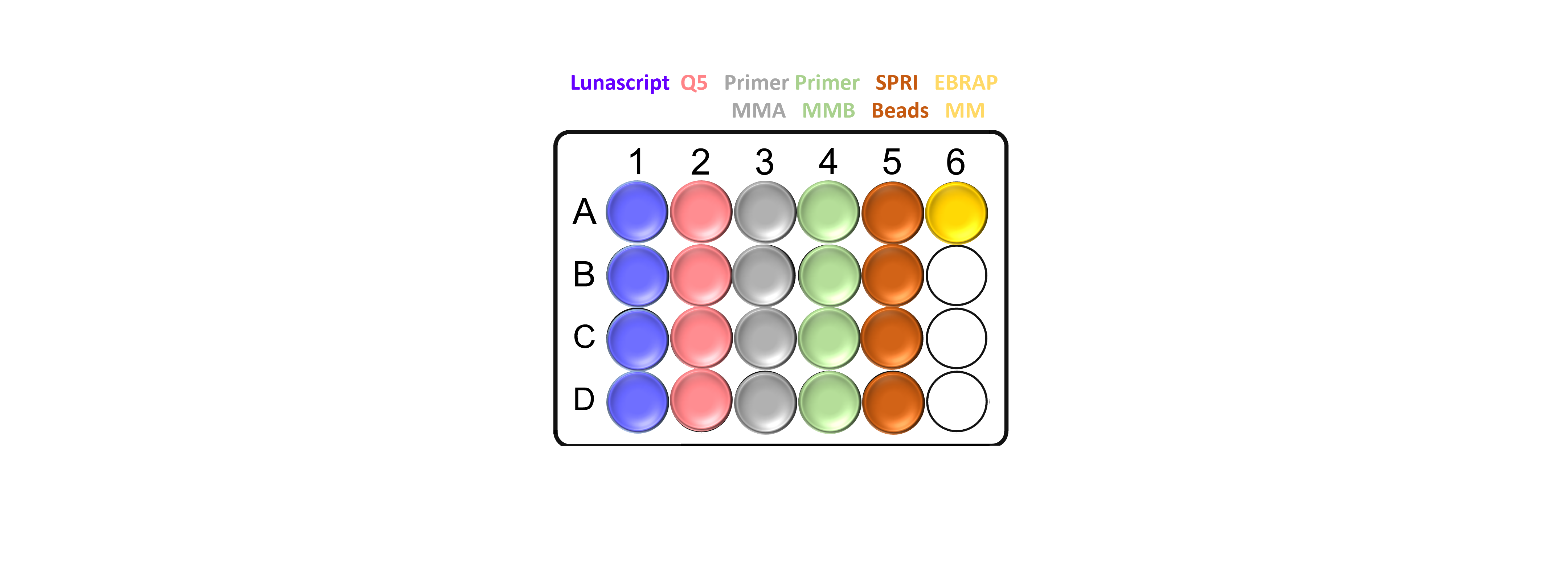
Note: It is important to throughly pipette mix all the reagents before aliquoting. Take care not to introduce air bubbles as this can cause errors during the automated process.
-
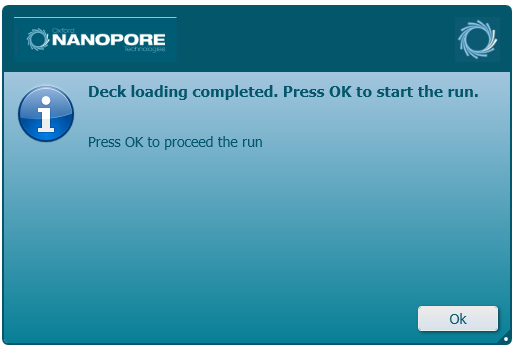
Once loading has been completed, click 'Ok' to being the run.
-
Once the cDNA Amplification PCR has been completed (on or off-deck), load the RBK plate (and the PCR plates if required) following the on-screen instructions. Click 'Ok' to continue the run.

-
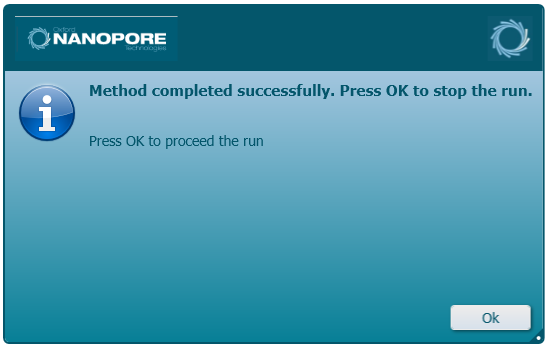
Upon successful run completion, the following message is shown. Click 'Ok' to continue.
-
Select and complete the required post-run steps. Click 'Ok' to continue.
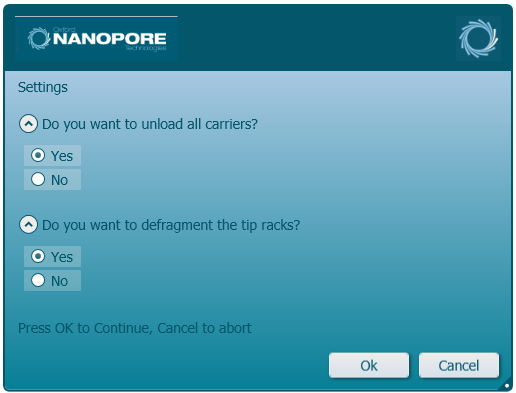
-
Perform the final checks and select 'OK' to complete the run.
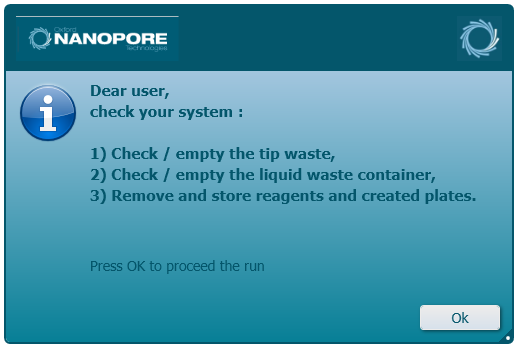
-
After the run ends, the final library can be collected from the designated position in the MIDI plate. Remove and retain the final elution into a clean 1.5 ml Eppendorf DNA LoBind tube.
-
Quantify DNA concentration of the final elution by using the Qubit dsDNA HS Assay Kit.

























