- Materials
-
- DNA CS
- 500 ng–1 µg captured DNA in 48 µl
- Consumables
-
- NEBNext Ultra II End repair/dA-tailing Module (NEB, E7546)
- Freshly prepared 70% ethanol in nuclease-free water
- 1.5 ml Eppendorf DNA LoBind tubes
- Nuclease-free water (e.g. ThermoFisher, AM9937)
- Agencourt AMPure XP beads (Beckman Coulter™ cat # A63881)
- Equipment
-
- Thermal cycler
- Magnetic rack, suitable for 1.5 ml Eppendorf tubes
- Hula mixer (gentle rotator mixer)
- Vortex mixer
- Ice bucket with ice
- Optional equipment
-
- Qubit fluorometer (or equivalent for QC check)
-
Perform end repair and dA-tailing as follows:
End-prep demo
Mix the following reagents in a 1.5 ml Eppendorf DNA LoBind tube:
Reagent Volume ~1 µg DNA (fragmented genomic DNA, amplicon or cDNA) 45 µl Ultra II End-prep reaction buffer 7 µl Ultra II End-prep enzyme mix 3 µl DNA CS 5 µl Total 60 µl If using the barcoding approach the pooled input DNA should be ~1 µg in 45 µl, whether genomic, amplicon or cDNA.
-
Mix gently by flicking the tube, and spin down.
-
Transfer the sample to a 0.2 ml PCR tube, and incubate for 5 minutes at 20° C and 5 minutes at 65° C using the thermal cycler.
If condensation is observed in the tube after the thermocycling, briefly spin down the tube contents in a microfuge.
-
Resuspend the AMPure XP beads by vortexing.
-
Transfer the sample to a clean 1.5 ml Eppendorf DNA LoBind tube.
-
Add 60 µl of resuspended AMPure XP beads to the end-prep reaction and mix by pipetting.
-
Incubate on a Hula mixer (rotator mixer) for 5 minutes at room temperature.
-
Prepare 500 μl of fresh 70% ethanol in nuclease-free water.
-
Spin down the sample and pellet on a magnet. Keep the tube on the magnet, and pipette off the supernatant when clear and colourless.
-
Keep the tube on the magnet and wash the beads with 200 µl of freshly prepared 70% ethanol without disturbing the pellet. Remove the ethanol using a pipette and discard.
-
Repeat the previous step.
-
Spin down and place the tube back on the magnet. Pipette off any residual ethanol. Allow to dry for ~30 seconds, but do not dry the pellet to the point of cracking.
-
Remove the tube from the magnetic rack and resuspend pellet in 31 µl nuclease-free water. Incubate for 2 minutes at room temperature.
-
Pellet the beads on a magnet until the eluate is clear and colourless.
-
Remove and retain 31 µl of eluate into a clean 1.5 ml Eppendorf DNA LoBind tube.
-
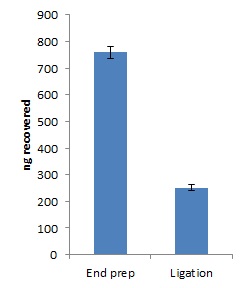
Quantify 1 µl of end-prepped DNA using a Qubit fluorometer - recovery aim >700 ng.