- Materials
-
- For Kit 14 libraries, Flow Cell Flush (FCF) and Flow Cell Tether (FCT)
- For Kit 9, 10 and 11 libraries, Flush Buffer (FB) and Flush Tether (FLT)
- Flow Cell Wash Kit (EXP-WSH004)
- Consumables
-
- Bovine Serum Albumin (BSA) (50 mg/ml) (e.g Invitrogen™ UltraPure™ BSA 50 mg/ml, AM2616)
- 1.5 ml Eppendorf DNA LoBind tubes
- Equipment
-
- MinION or GridION device
- P1000 pipette and tips
- P200 pipette and tips
- Vortex mixer
- Microfuge
- Ice bucket with ice
-
Preparation to recover and wash a library to replace on the same flow cell
- This protocol assumes that the original flow cell has been loaded with a DNA library prepared in accordance with the suitable protocol.
- The aim is to recover the library and wash the flow cell to reload the recovered library.
- Data acquisition in MinKNOW can be paused during the procedure.
-
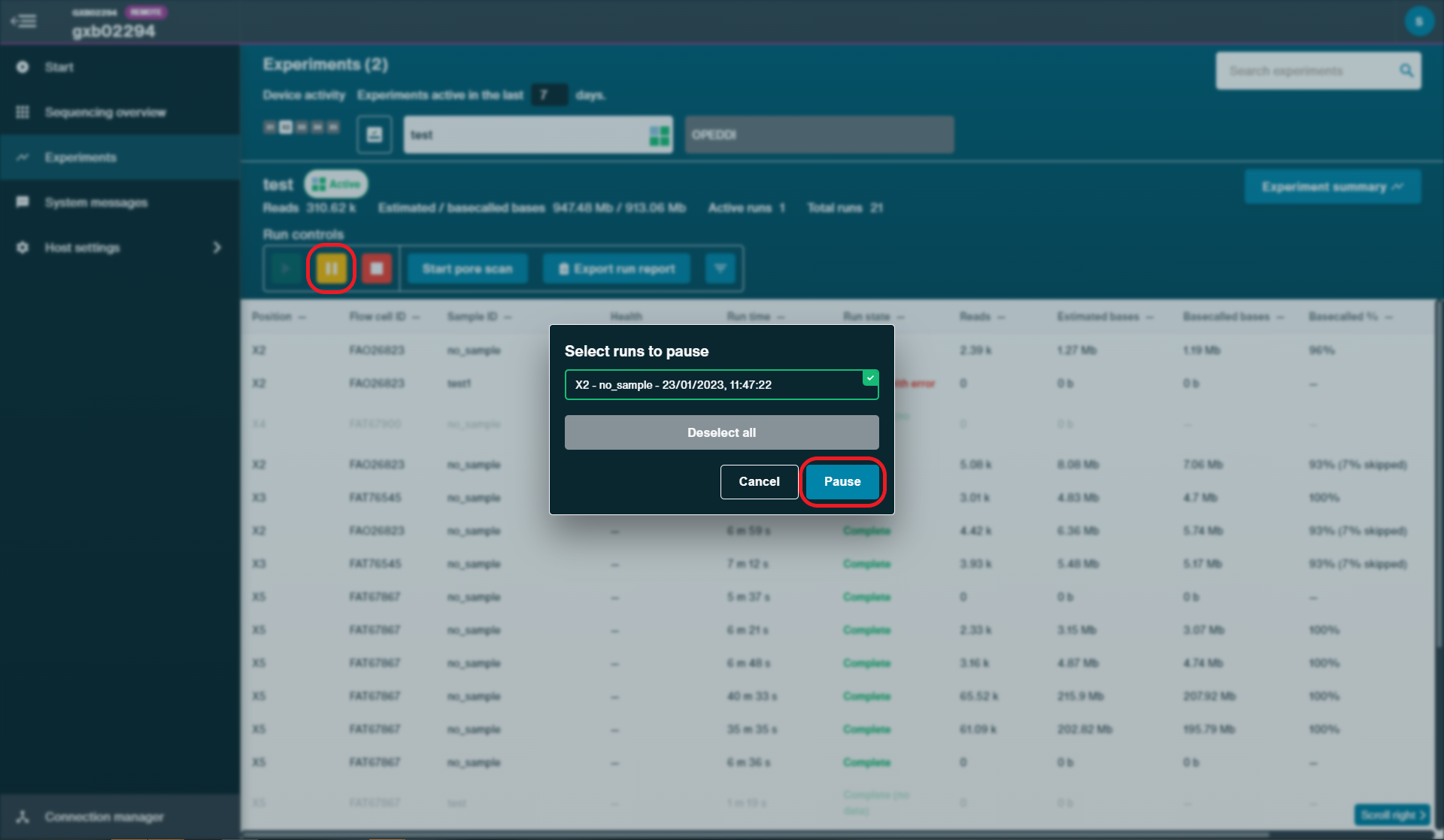
Pause the sequencing run for the original flow cell on MinKNOW by clicking 'Pause'.
-
Place the tube of Wash Mix (WMX) on ice. Do not vortex the tube.
-
Thaw one tube of Wash Diluent (DIL) at room temperature and mix the contents of Wash Diluent (DIL) thoroughly by vortexing. Then spin down briefly and place on ice.
-
To prepare the original flow cell for library recovery, slide open the priming port cover.
-
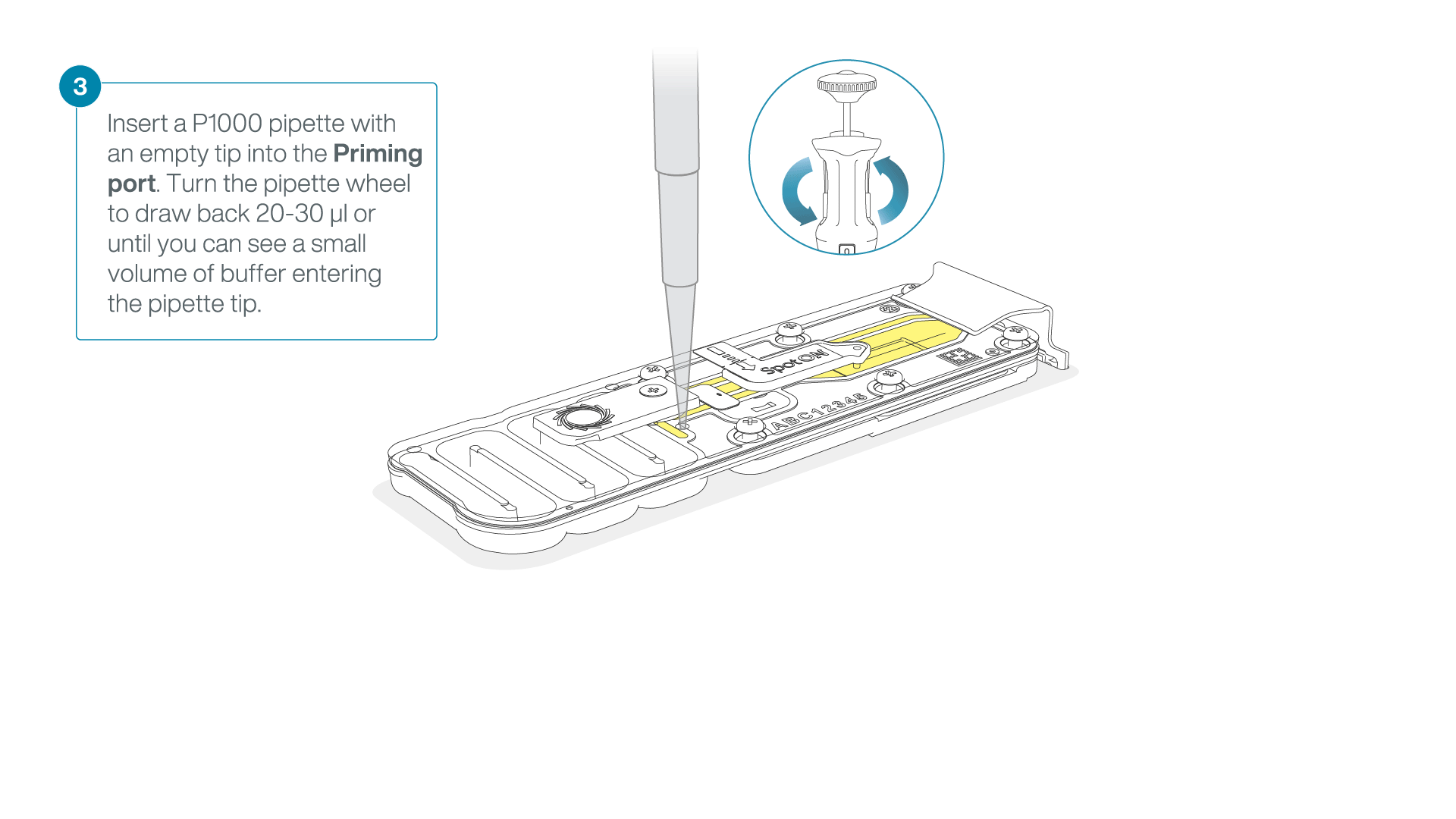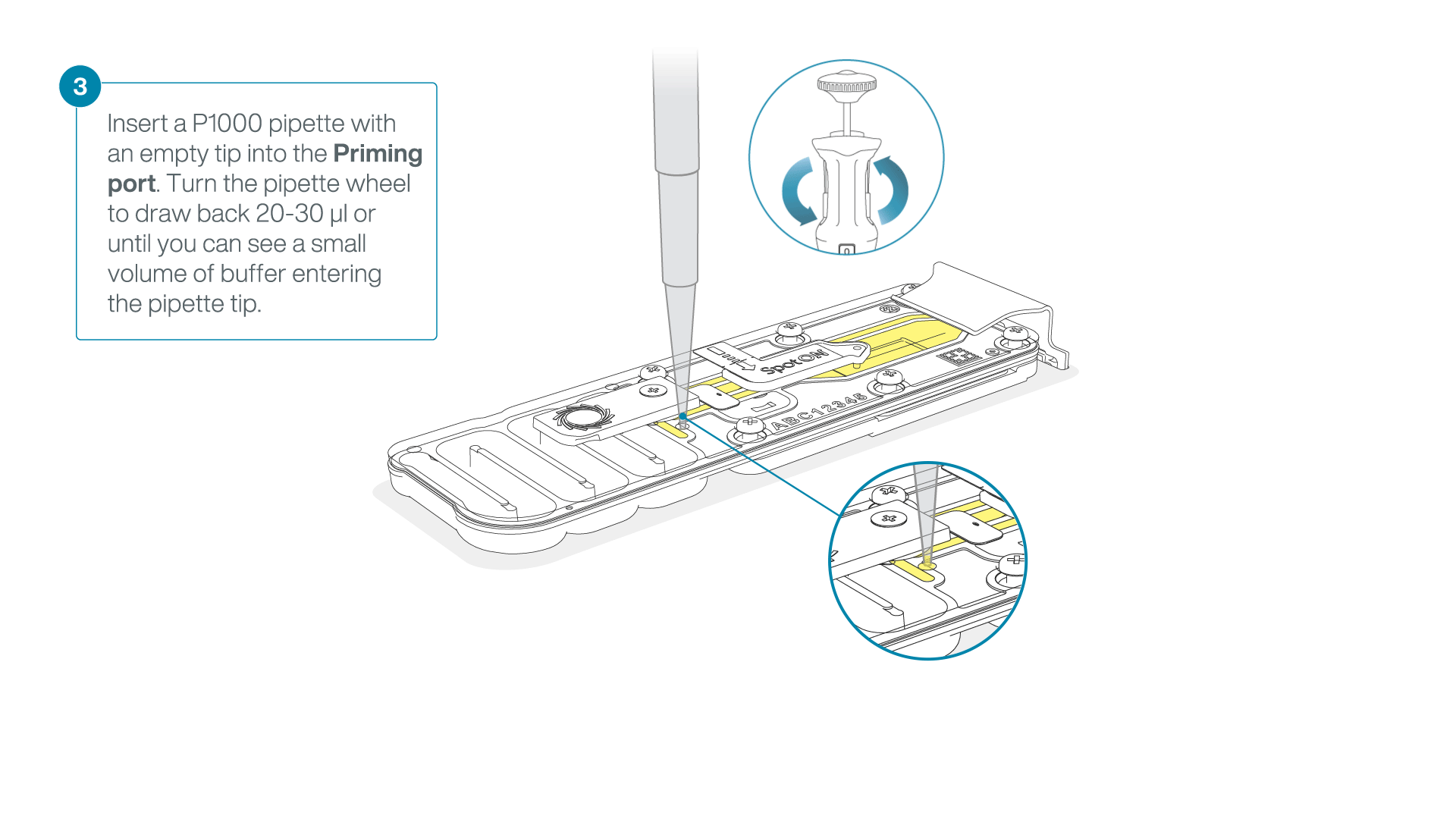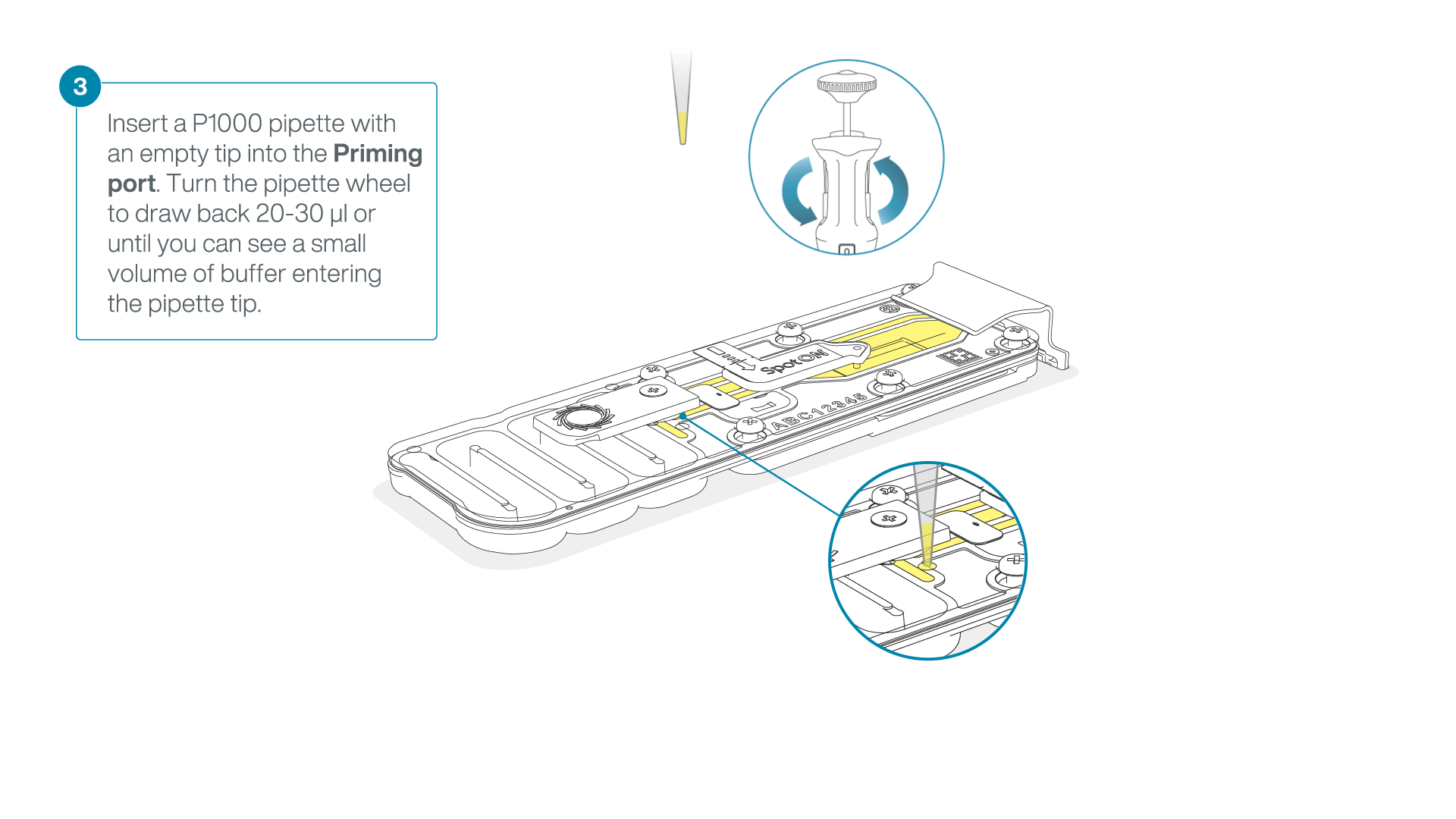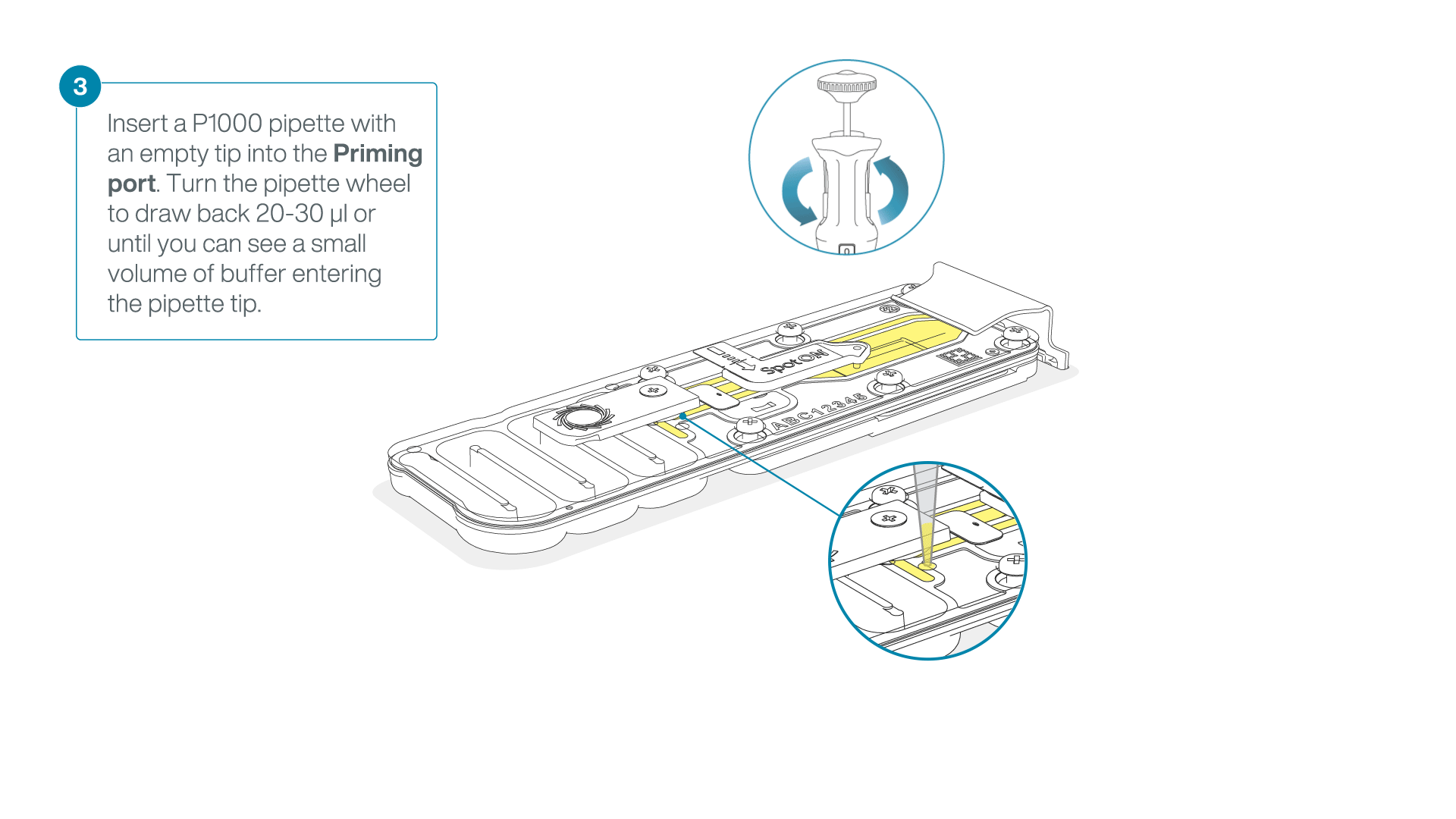
After opening the priming port, check for a small air bubble under the cover. Draw back a small volume to remove any bubbles:
- Set a P1000 pipette to 200 µl.
- Insert the tip into the flow cell priming port.
- Turn the wheel until the dial shows 220-230 µl, or until you can see a small volume of buffer/liquid entering the pipette tip.
- Visually check that there is continuous buffer from the flow cell priming port across the sensor array.
-
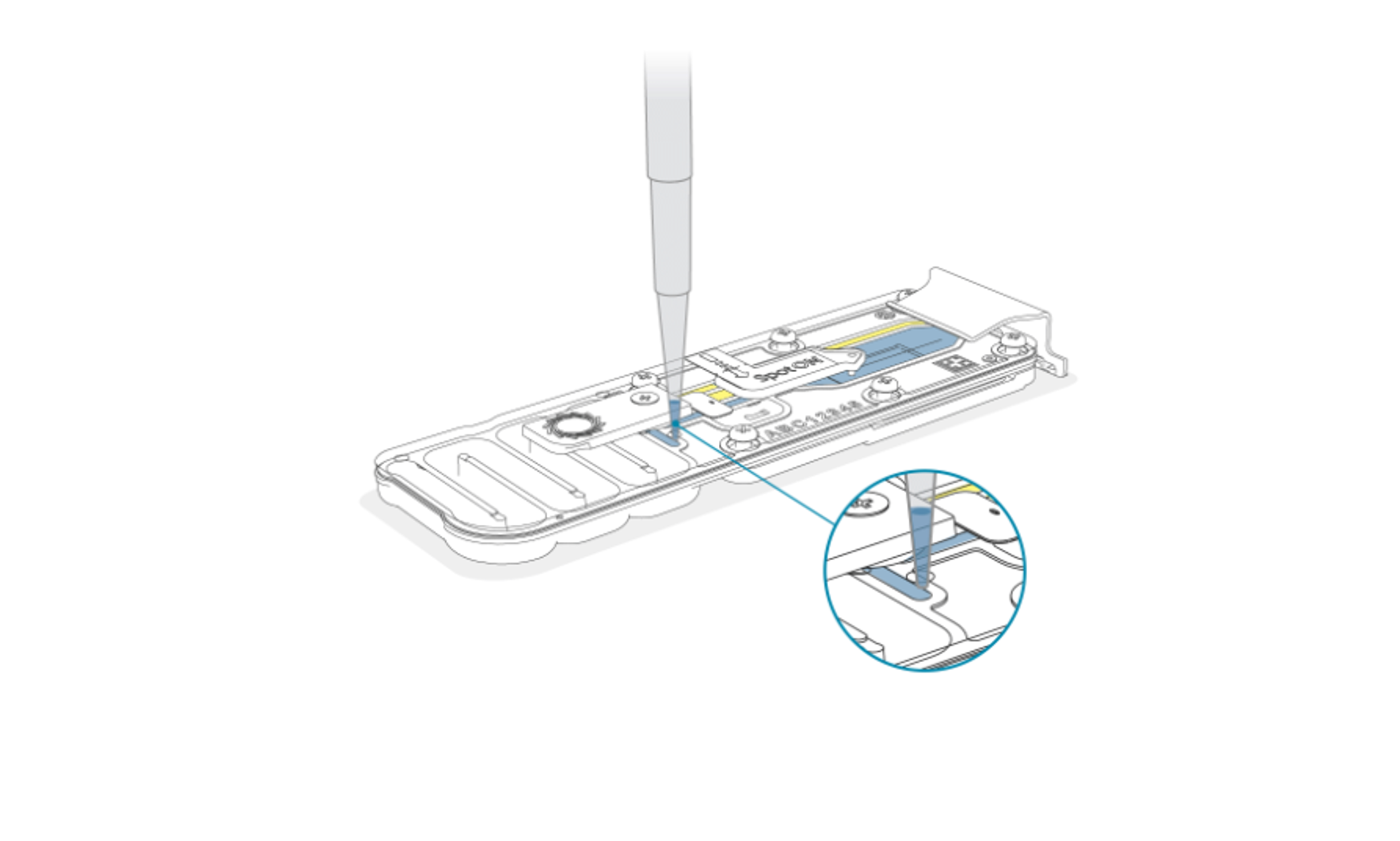
Set a pipette to 150 µl and fully depress the pipette before inserting the tip into the priming port of the original flow cell. Slowly aspirate to recover the DNA library from the flow cell.
Note: The recovered library may appear slightly yellow and not all the library loading beads (LB, LBII, LIB) will be fully recovered but this will not impact library recovery and transfer.
-
Transfer the recovered library to a fresh 1.5 ml Eppendorf DNA LoBind tube and store on ice.
-
In a fresh 1.5 ml Eppendorf DNA LoBind tube, prepare the following Flow Cell Wash Mix:
Reagent Volume per flow cell Wash Mix (WMX) 2 μl Wash Diluent (DIL) 398 μl Total 400 μl -
Mix well by pipetting, and place on ice. Do not vortex the tube.
-
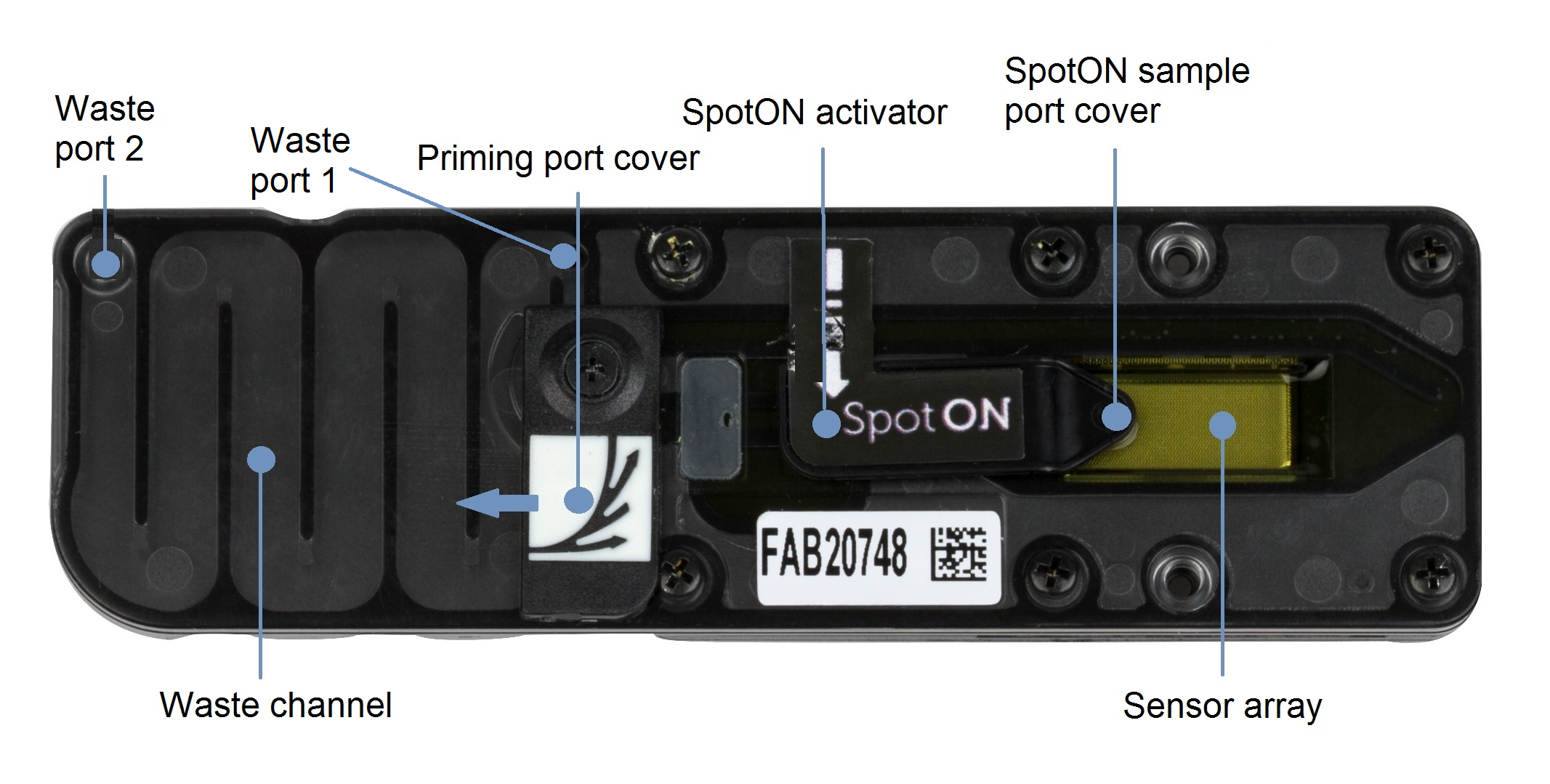
Remove the waste buffer, as follows:
- Close the priming port and SpotON sample port cover, as indicated in the figure below.
- Insert a P1000 pipette into waste port 1 and remove the waste buffer.
Note: As both the priming port and SpotON sample port are closed, no fluid should leave the sensor array area.
-
Rotate the flow cell priming port cover clockwise so that the priming port is visible.
-
After opening the priming port, check for a small air bubble under the cover. Draw back a small volume to remove any bubbles:
- Set a P1000 pipette to 200 µl.
- Insert the tip into the flow cell priming port.
- Turn the wheel until the dial shows 220-230 µl, or until you can see a small volume of buffer/liquid entering the pipette tip.
- Visually check that there is continuous buffer from the flow cell priming port across the sensor array.

-
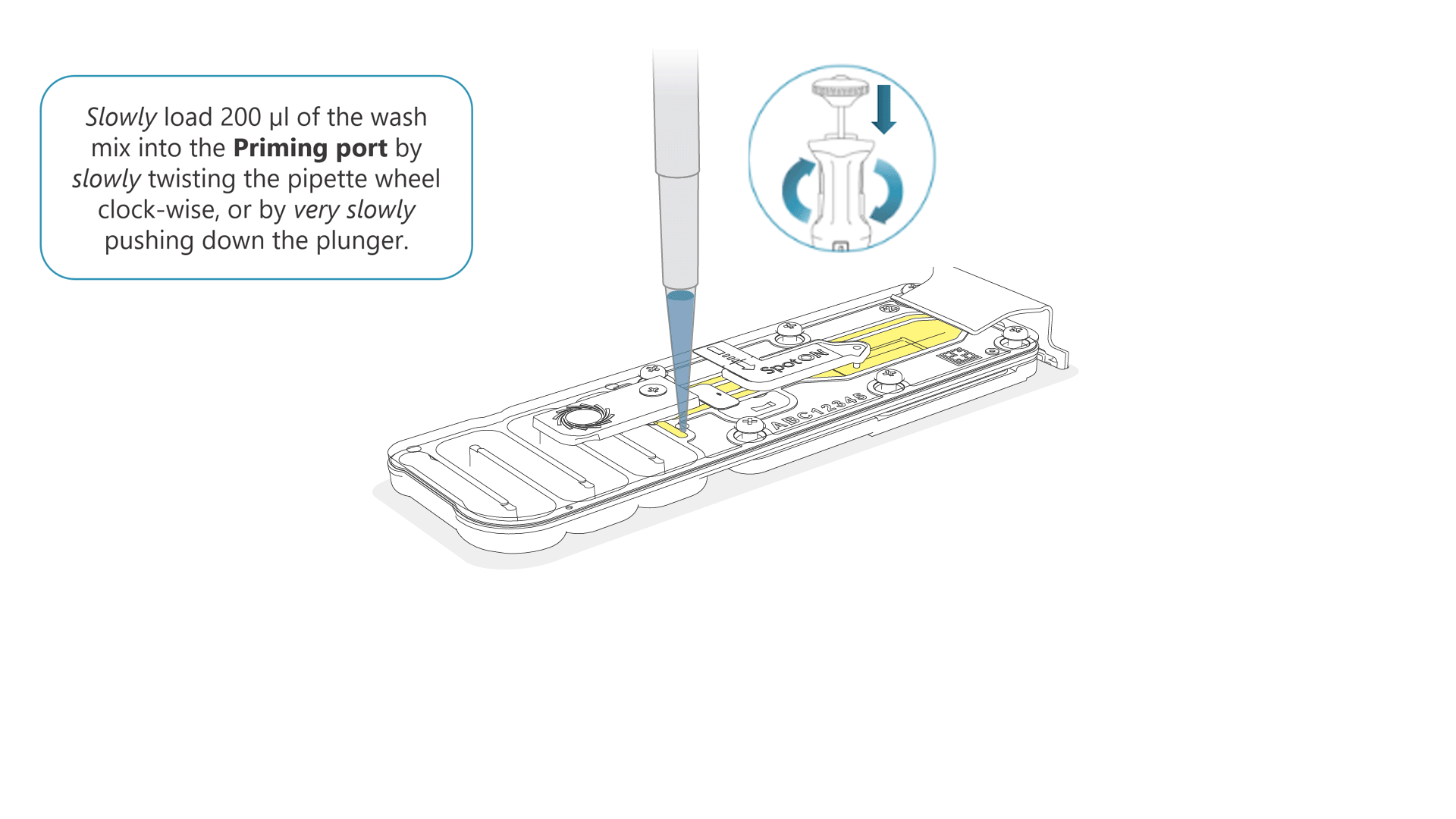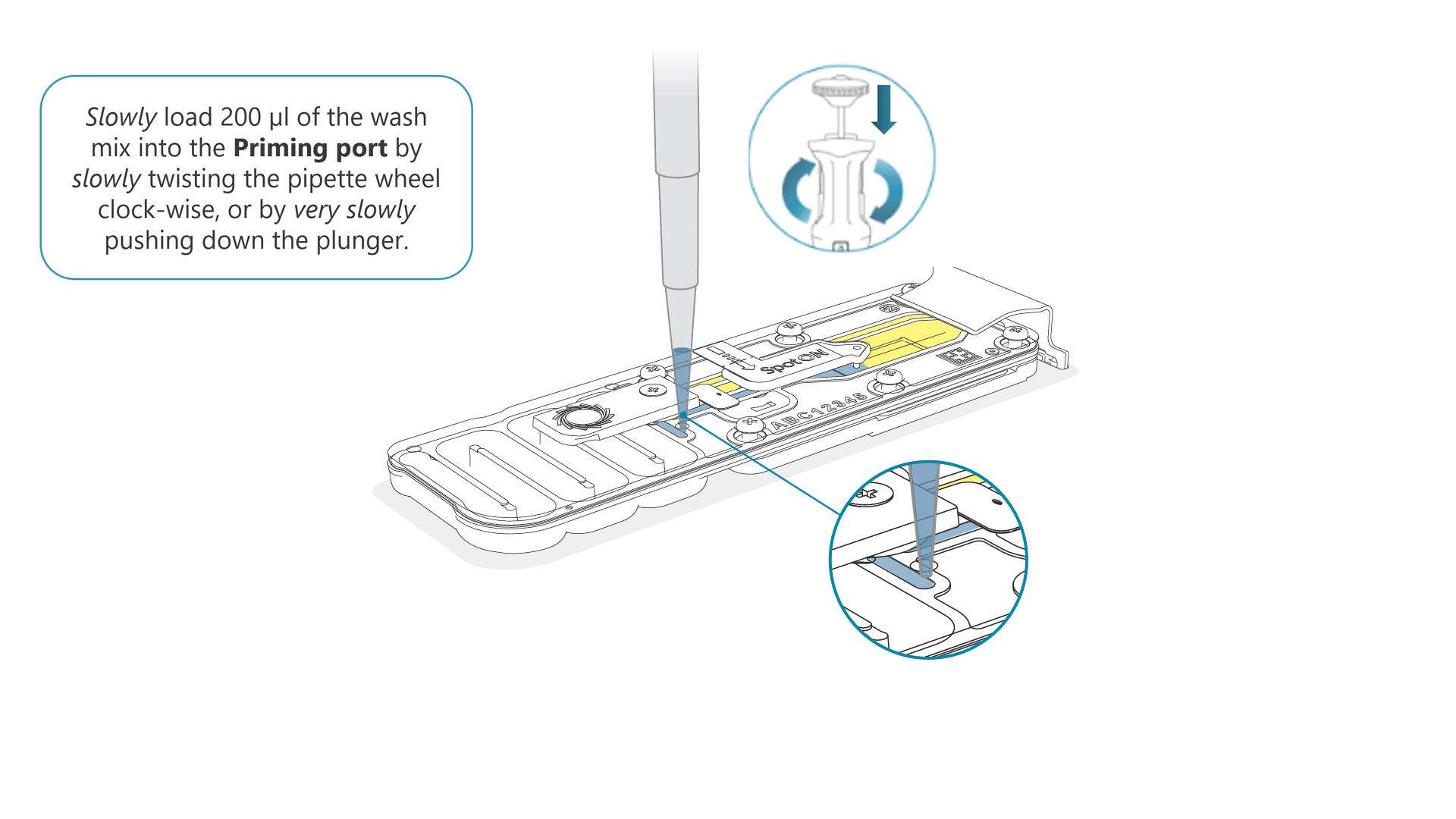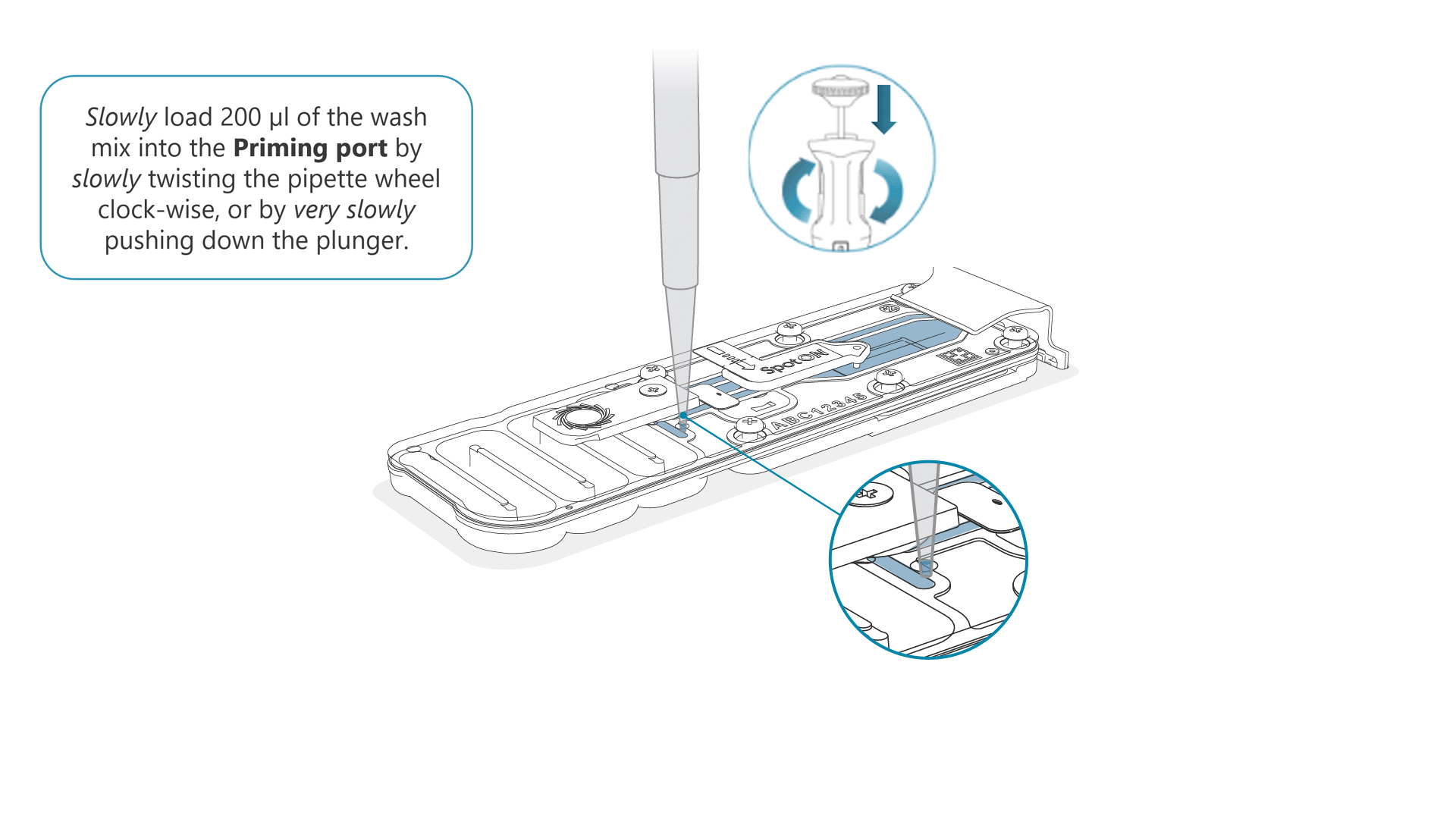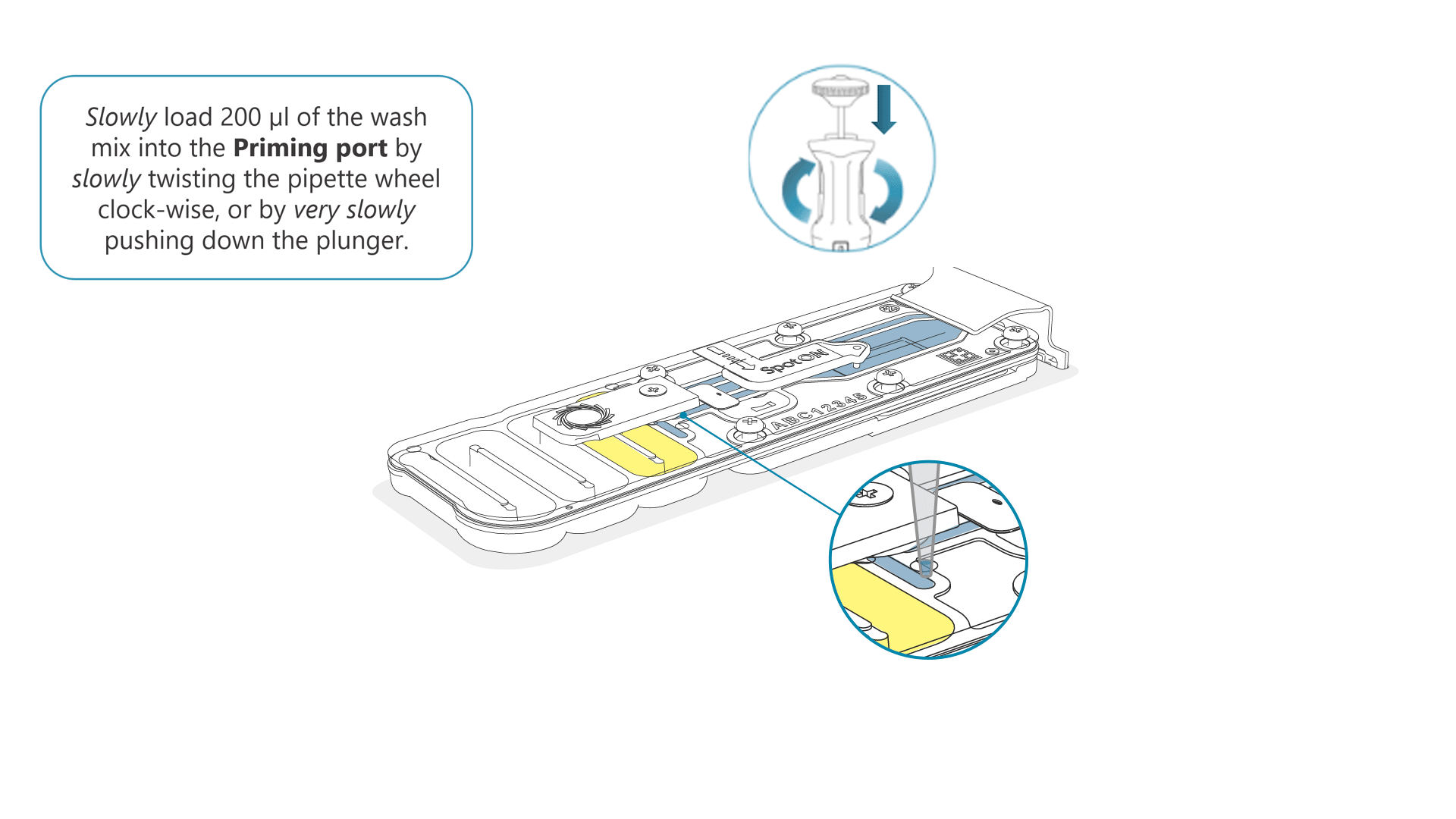
Slowly load 200 µl of the prepared flow cell wash mix into the priming port, as follows:
- Using a P1000 pipette, take 200 µl of the flow cell wash mix
- Insert the pipette tip into the priming port, ensuring there are no bubbles in the tip
- Slowly twist the pipette wheel down to load the flow cell (if possible with your pipette) or push down the plunger very slowly, leaving a small volume of buffer in the pipette tip.
- Set a timer for a 5 minute incubation.
-
Once the 5 minute incubation is complete, carefully load the remaining 200 µl of the prepared flow cell wash mix into the priming port, as follows:
- Using a P1000 pipette, take the remaining 200 µl of the flow cell wash mix
- Insert the pipette tip into the priming port, ensuring there are no bubbles in the tip
- Slowly twist the pipette wheel down to load the flow cell (if possible with your pipette) or push down the plunger very slowly, leaving a small volume of buffer in the pipette tip.

-
Close the priming port and wait for 1 hour.
-
Remove the waste buffer, as follows:
- Close the priming port and SpotON sample port cover, as indicated in the figure below.
- Insert a P1000 pipette into waste port 1 and remove the waste buffer.
Note: As both the priming port and SpotON sample port are closed, no fluid should leave the sensor array area.

-
Thaw and prepare the flow cell priming mix according to the "Priming and loading the SpotON flow cell" section of the suitable protocol.
-
Slide the flow cell priming port cover clockwise to open the priming port.
-
After opening the priming port, check for a small air bubble under the cover. Draw back a small volume to remove any bubbles:
- Set a P1000 pipette to 200 µl
- Insert the tip into the priming port
- Turn the wheel until the dial shows 220-230 µl, to draw back 20-30 µl, or until you can see a small volume of buffer entering the pipette tip
Note: Visually check that there is continuous buffer from the priming port across the sensor array.

-
Load 800 µl of the priming mix into the flow cell via the priming port, avoiding the introduction of air bubbles. Wait for five minutes.
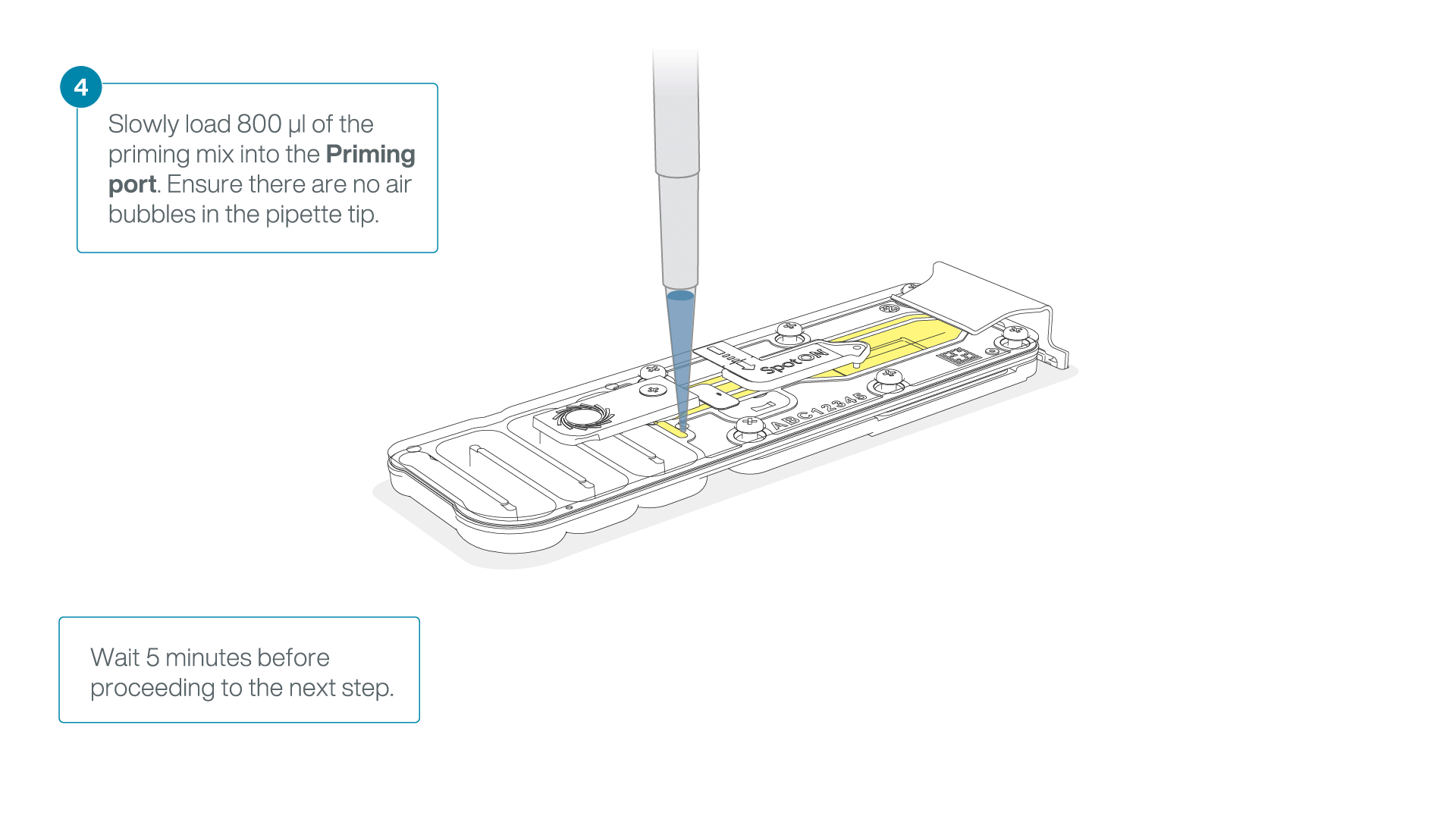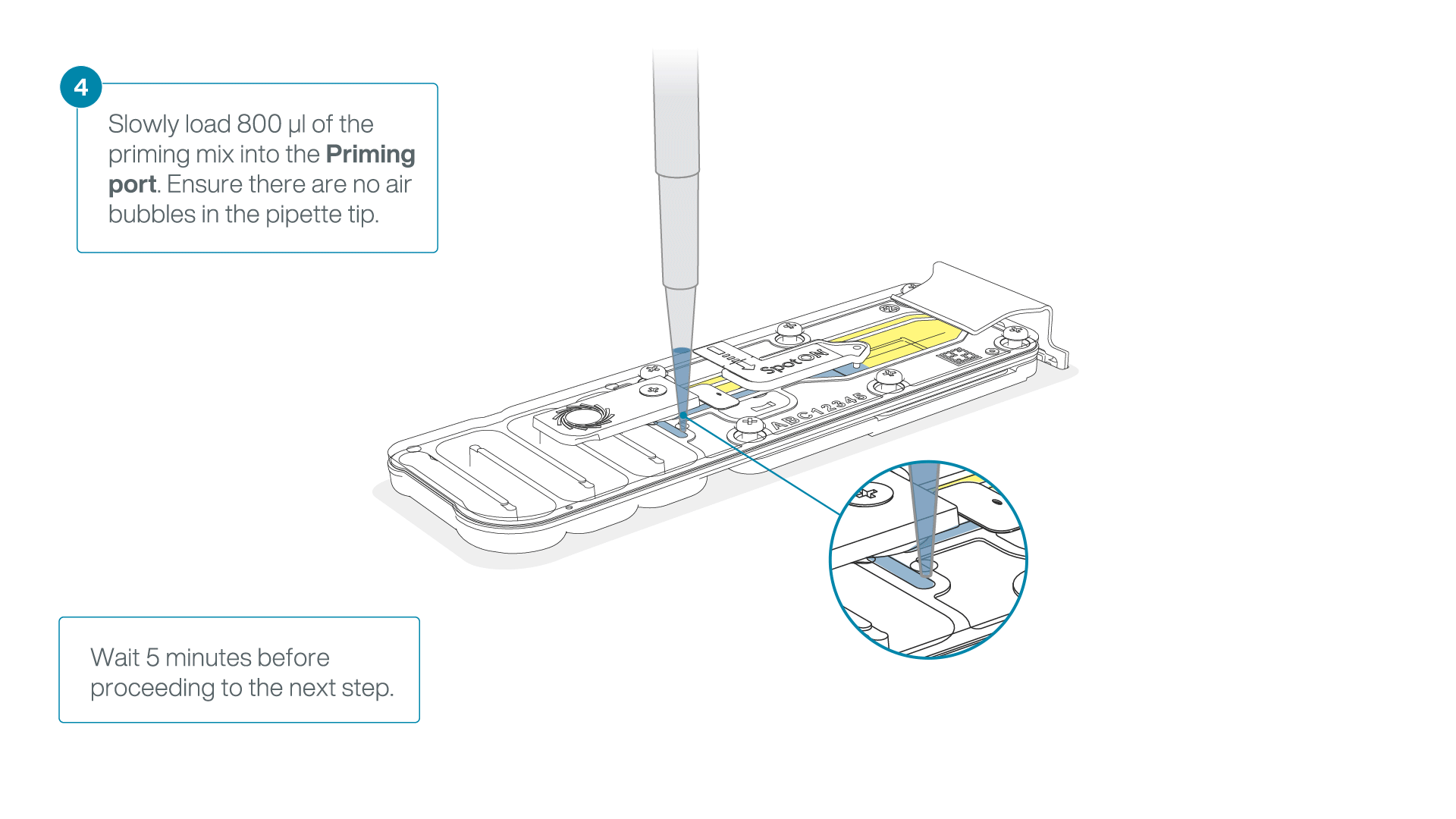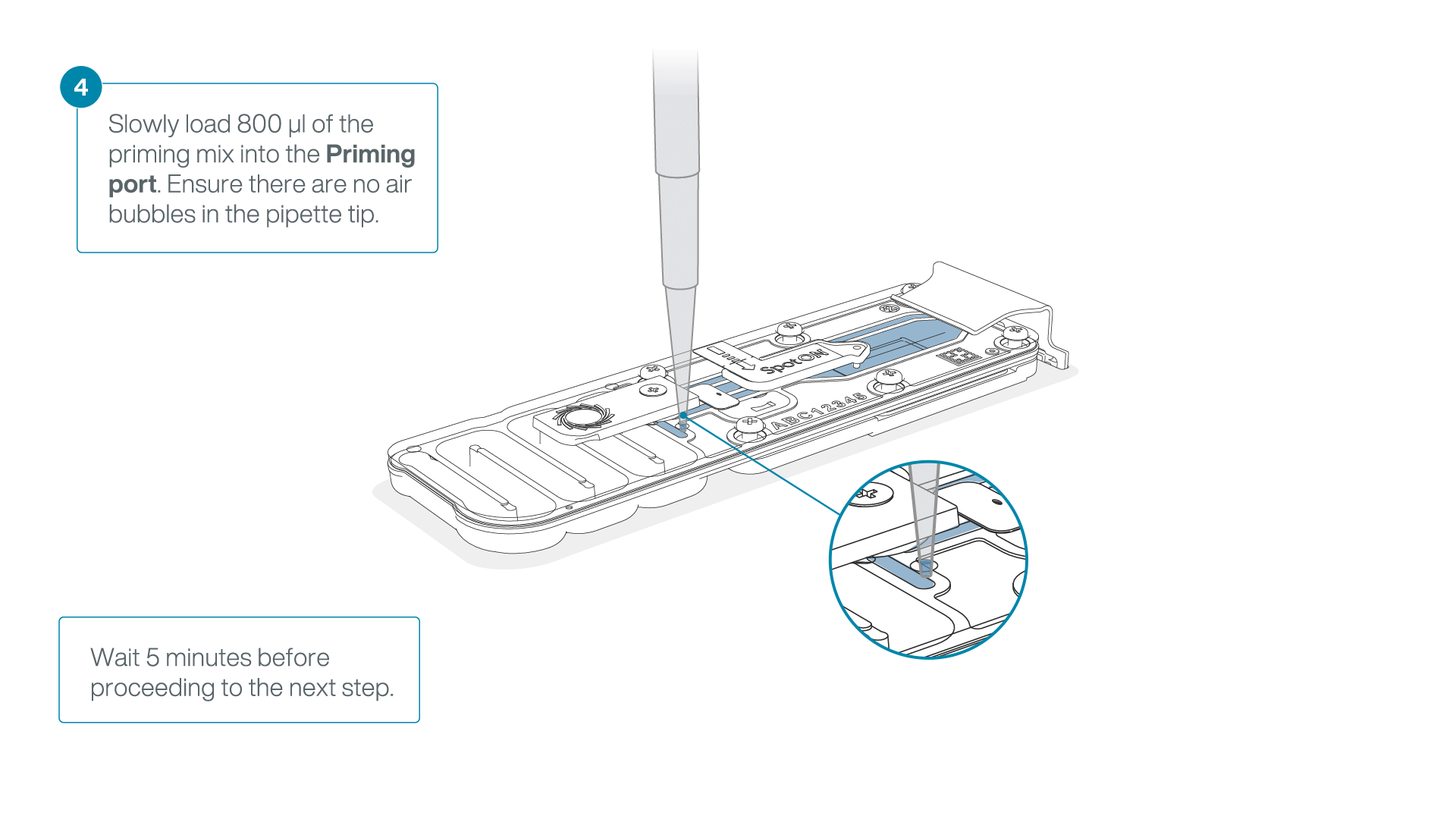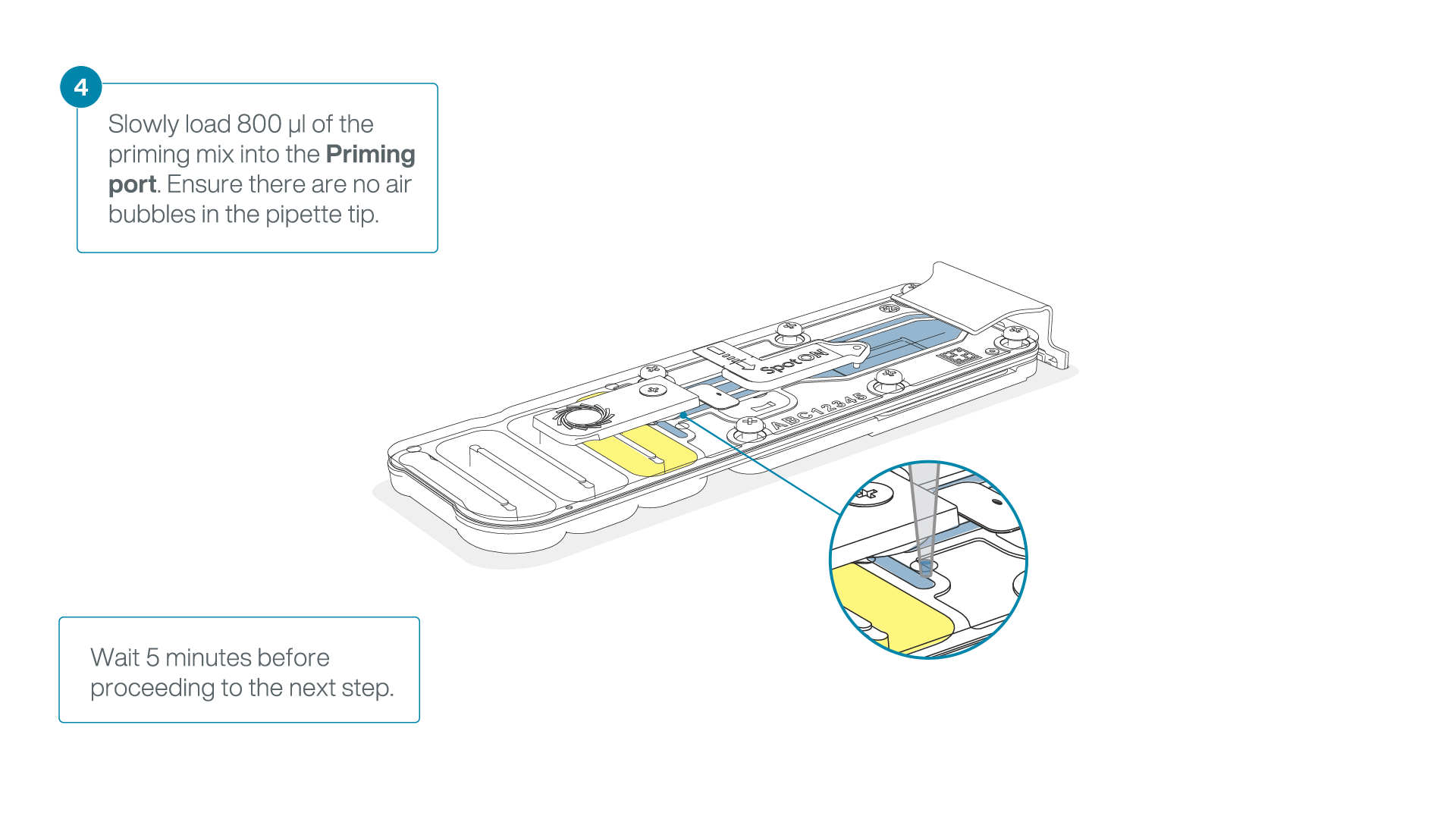
-
Complete the flow cell priming:
- Gently lift the SpotON sample port cover to make the SpotON sample port accessible.
- Load 200 µl of the priming mix into the flow cell via the priming port (not the SpotON sample port), avoiding the introduction of air bubbles.
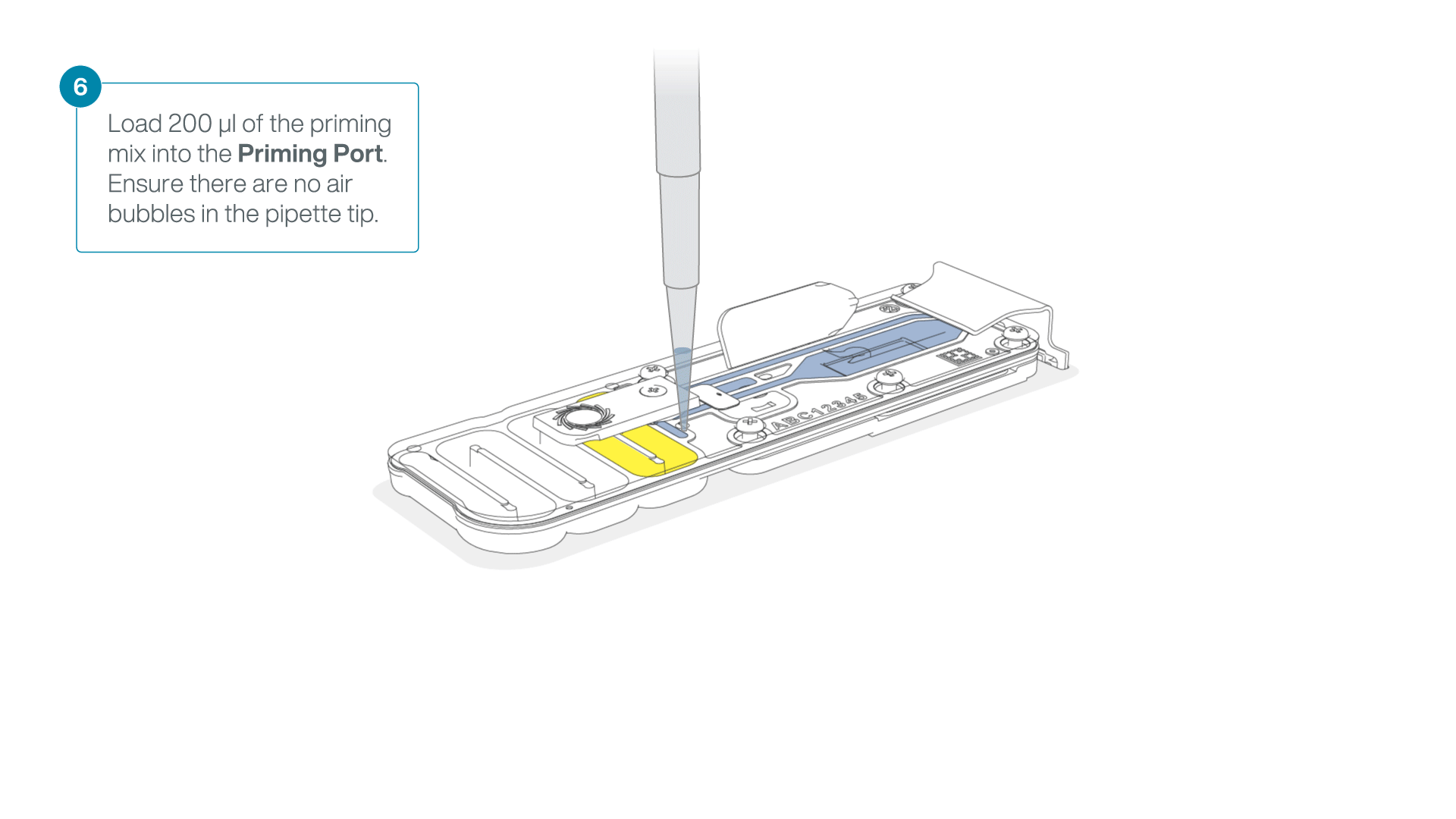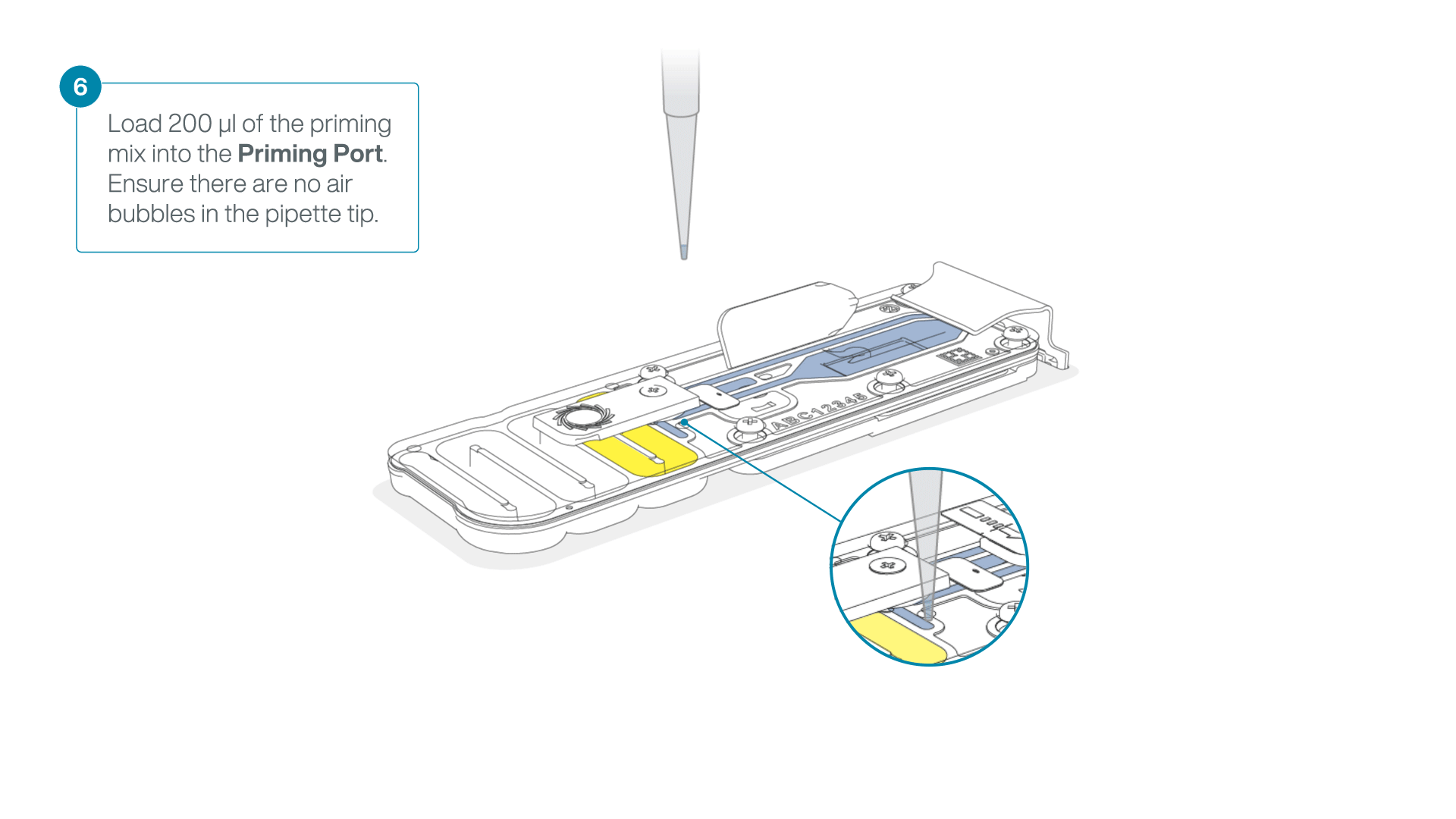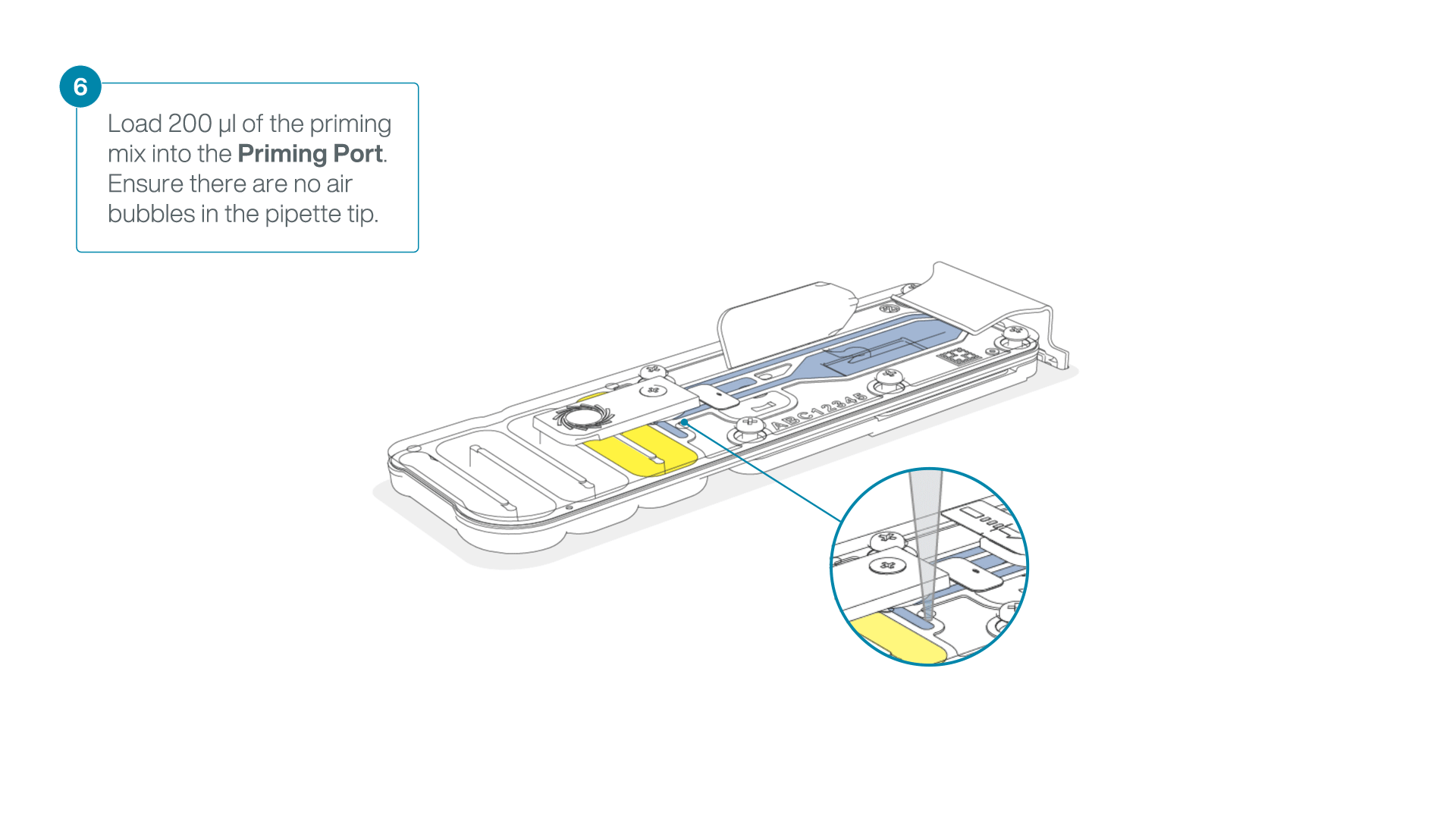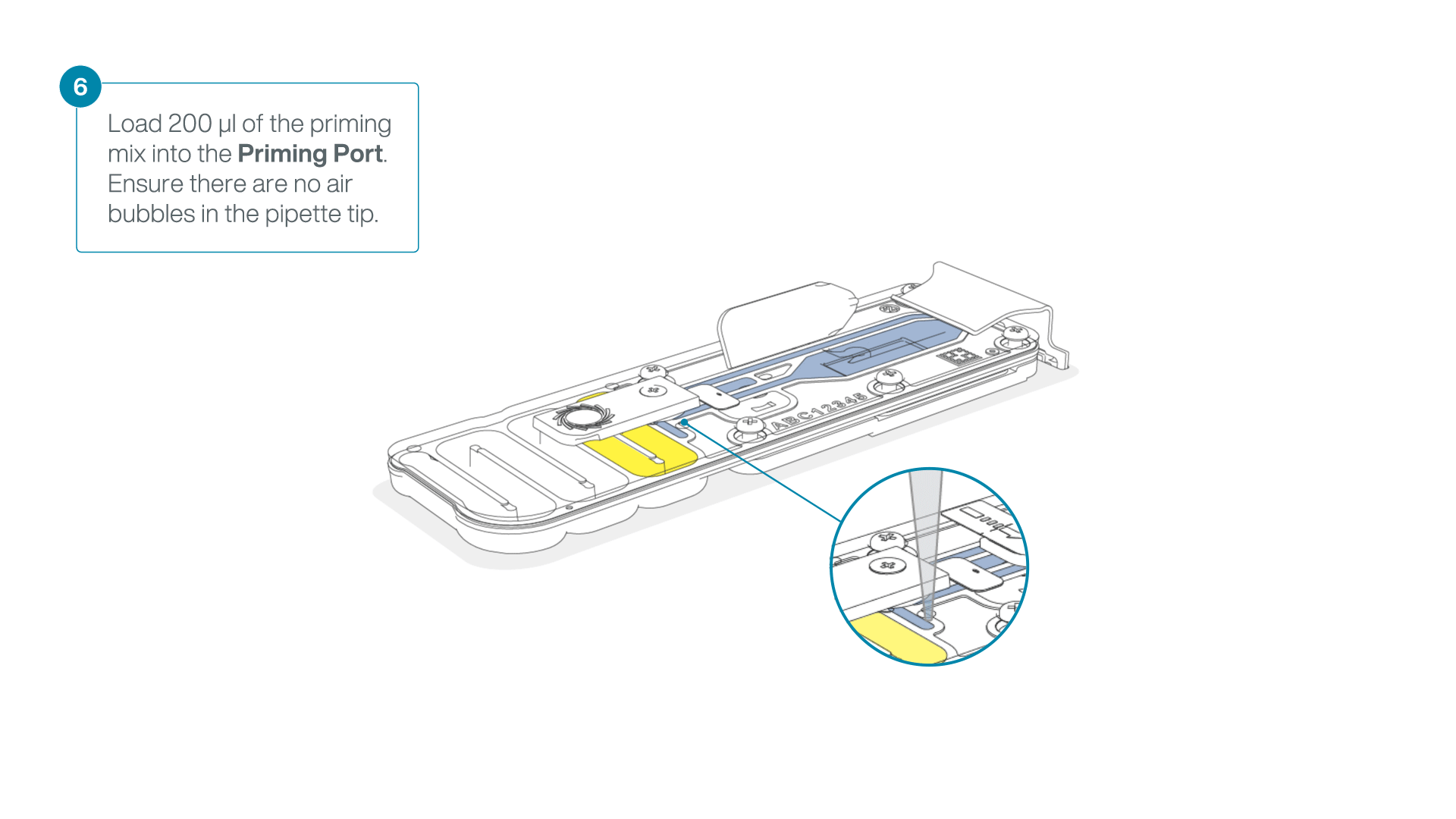
-
Bring the recovered library up to room temperature and load 150 µl to the flow cell via the SpotON port in a dropwise fashion. Ensure each drop flows into the port before adding the next.
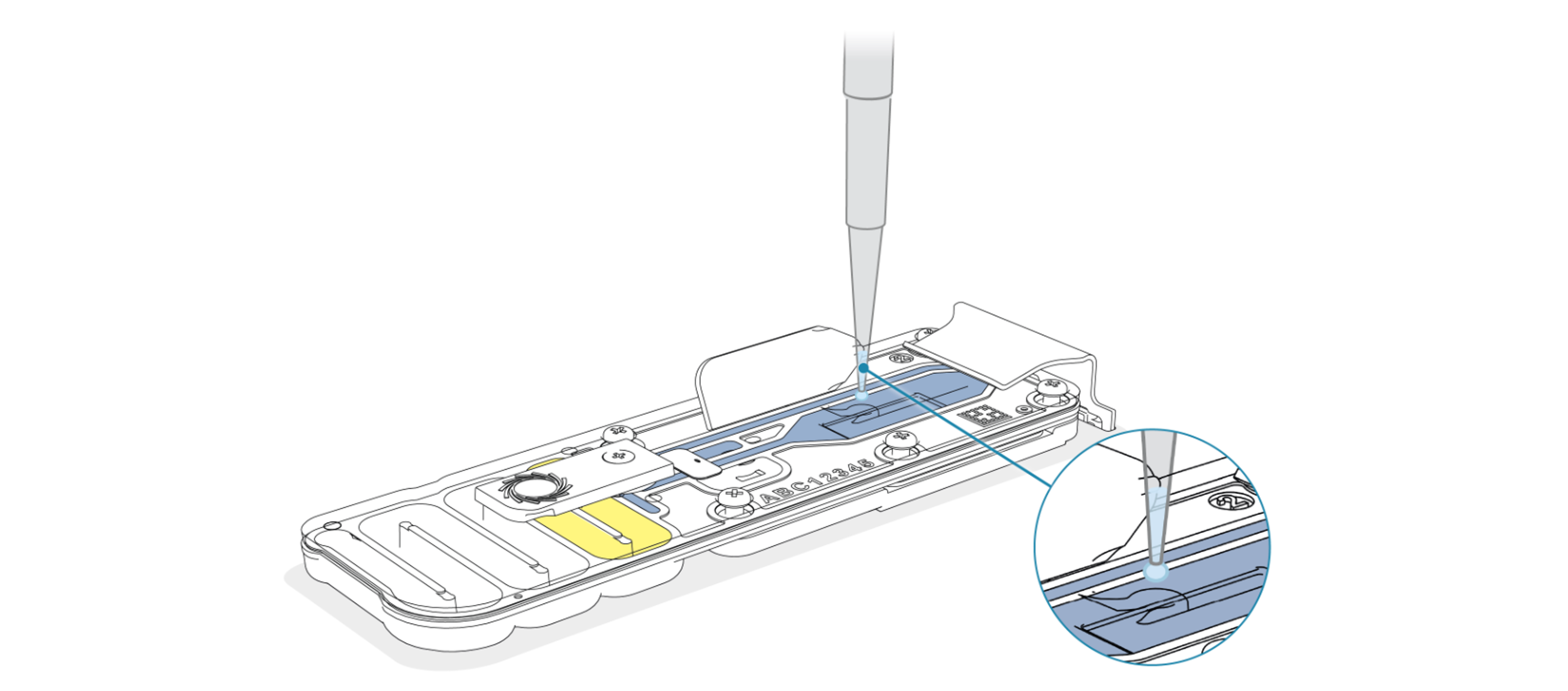
-
Gently replace the SpotON sample port cover, making sure the bung enters the SpotON port and close the priming port.

-
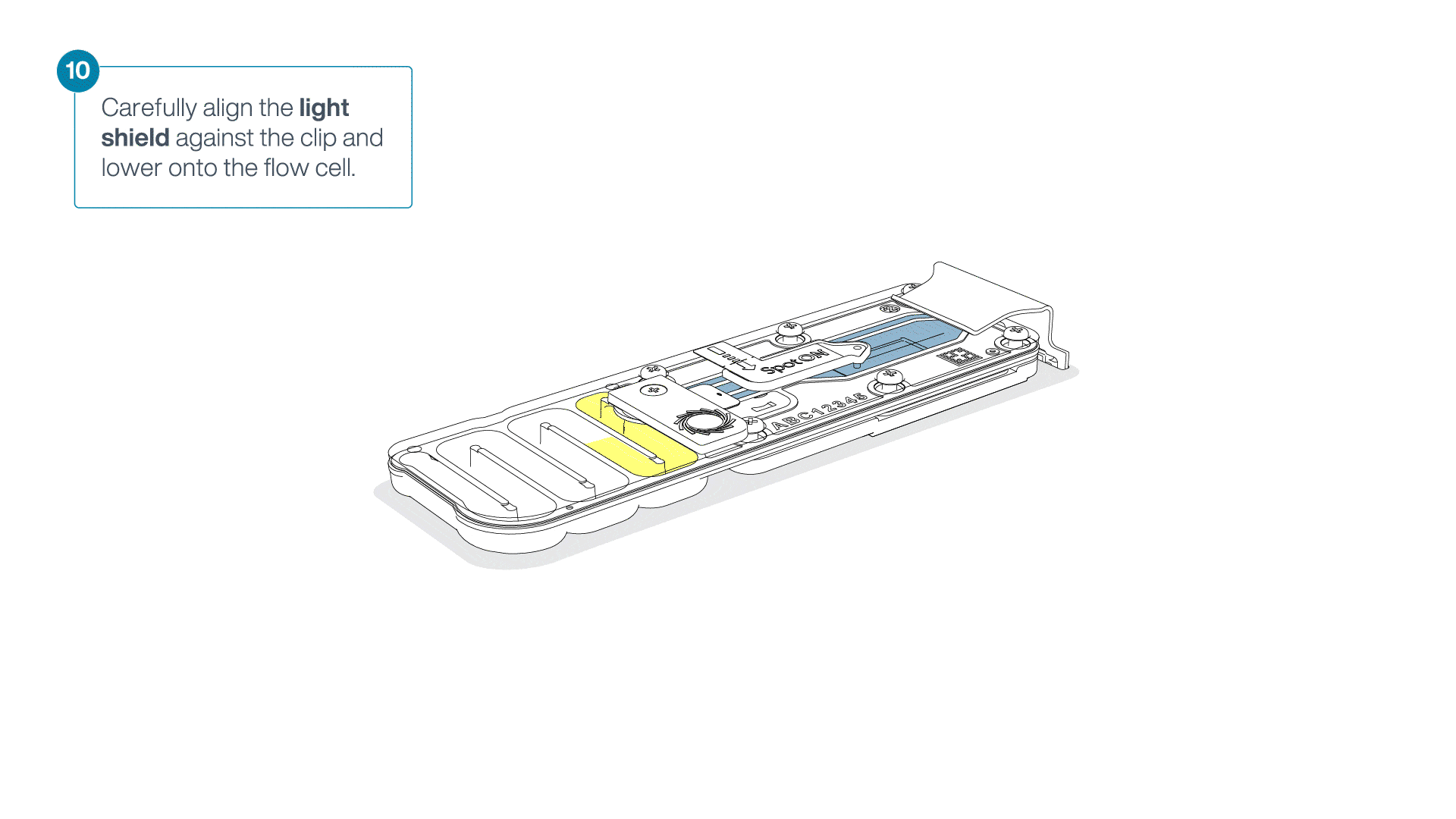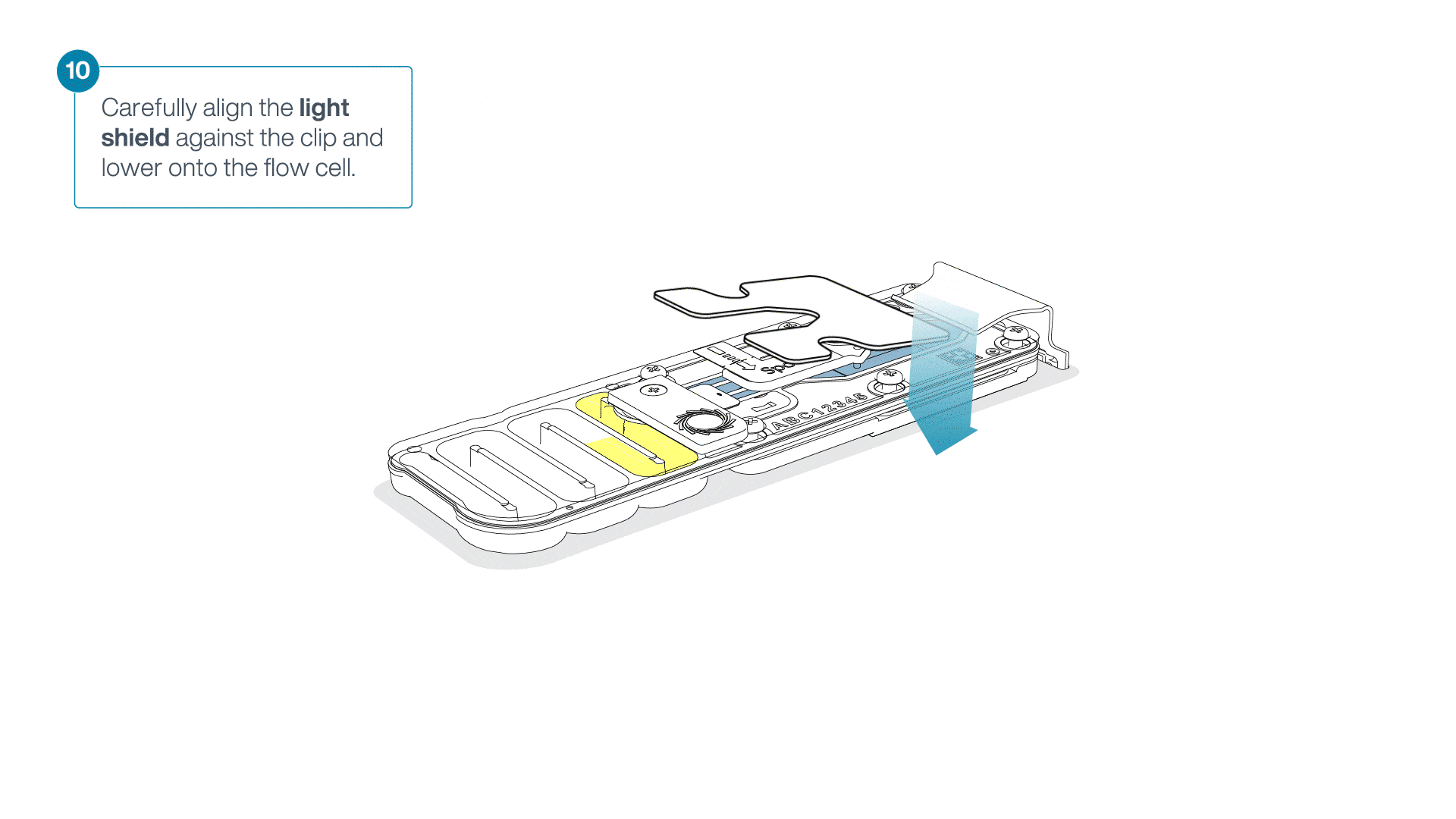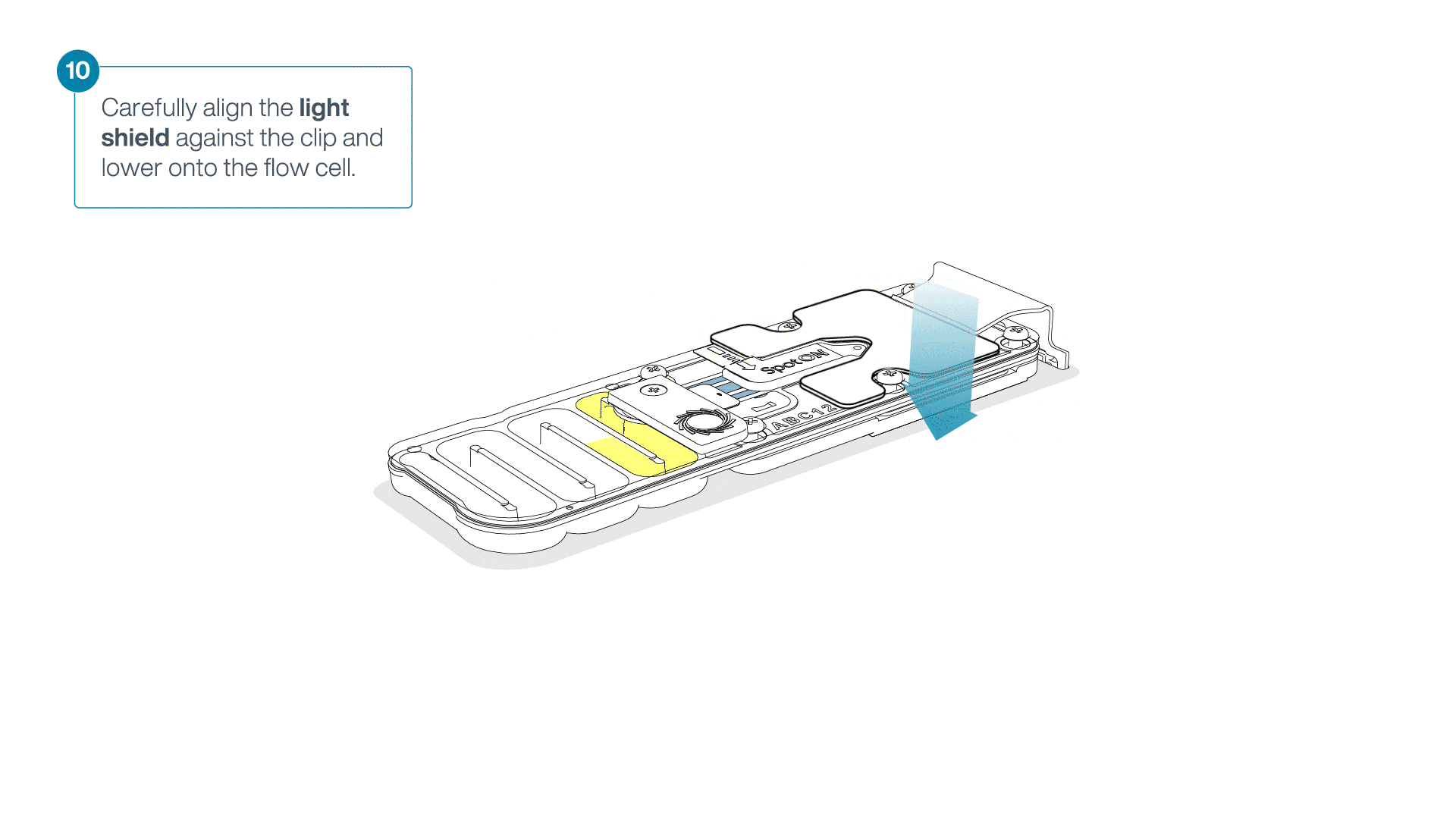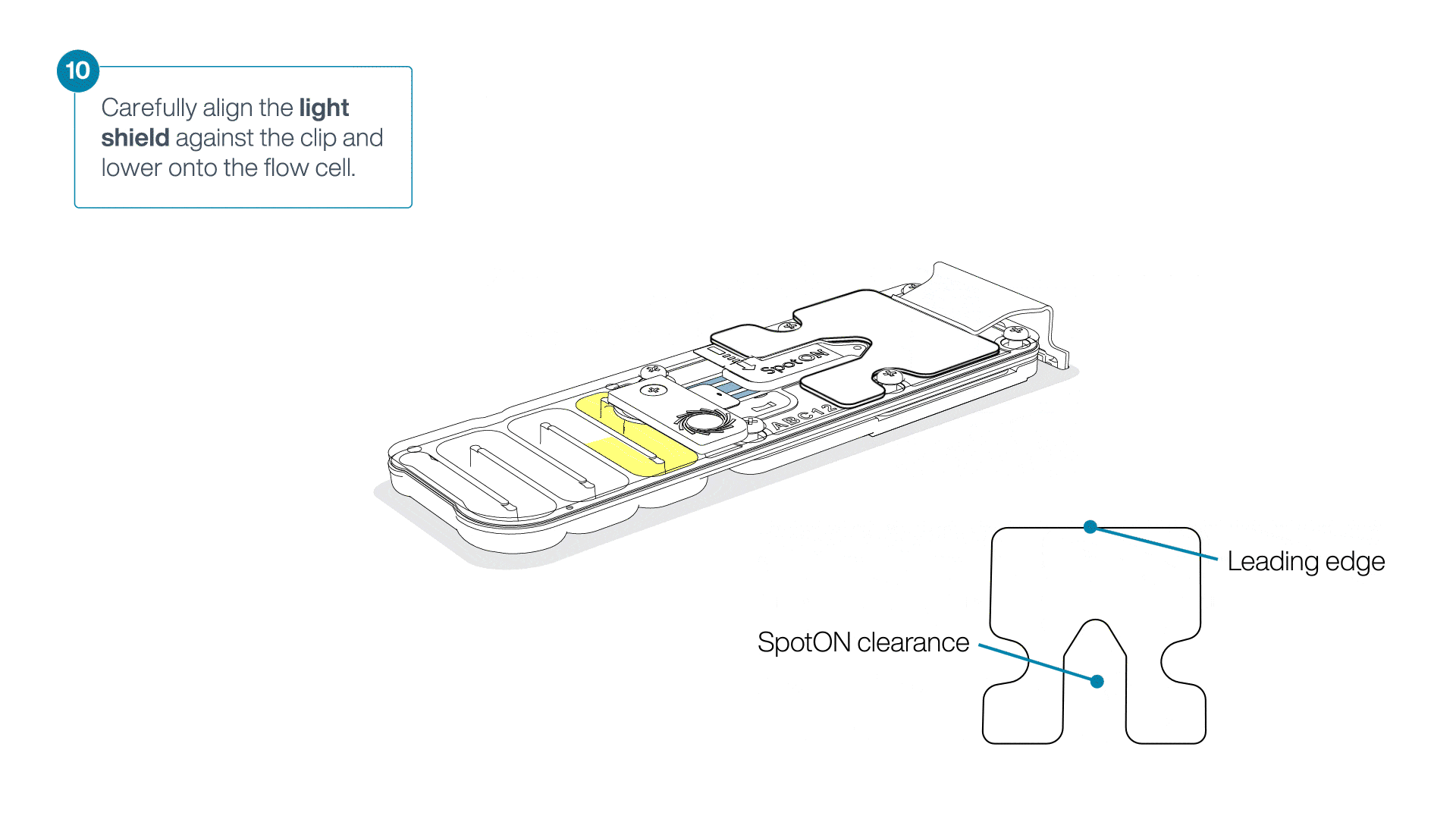
Place the light shield onto the flow cell, as follows:
Carefully place the leading edge of the light shield against the clip.
Note: Do not force the light shield underneath the clip.Gently lower the light shield onto the flow cell. The light shield should sit around the SpotON cover, covering the entire top section of the flow cell.
-
Restart the sequencing run on MinKNOW.









