- Materials
-
- For Kit 14 libraries, Flow Cell Flush (FCF) and Flow Cell Tether (FCT)
- For Kit 9, 10 and 11 libraries, Flush Buffer (FB) and Flush Tether (FLT)
- Consumables
-
- MinION Flow Cell (FLO-MIN106 or FLO-MIN114)
- Bovine Serum Albumin (BSA) (50 mg/ml) (e.g Invitrogen™ UltraPure™ BSA 50 mg/ml, AM2616)
- Equipment
-
- MinION or GridION device
- MinION and GridION Flow Cell Light Shield
- P1000 pipette and tips
- P200 pipette and tips
-
Preparation to transfer a library to a second flow cell
- This protocol assumes that the original flow cell has been loaded with a DNA library prepared in accordance with the suitable protocol.
- The aim is to prepare the second flow cell and recover the library from the original flow cell for immediate transfer.
- Data acquisition in MinKNOW should be stopped during the recovery and transfer procedure.
- After the second flow cell has been primed, the library can be recovered and immediately transferred to the second flow cell.
-
Stop the sequencing run for the original flow cell on MinKNOW by clicking 'Stop'.
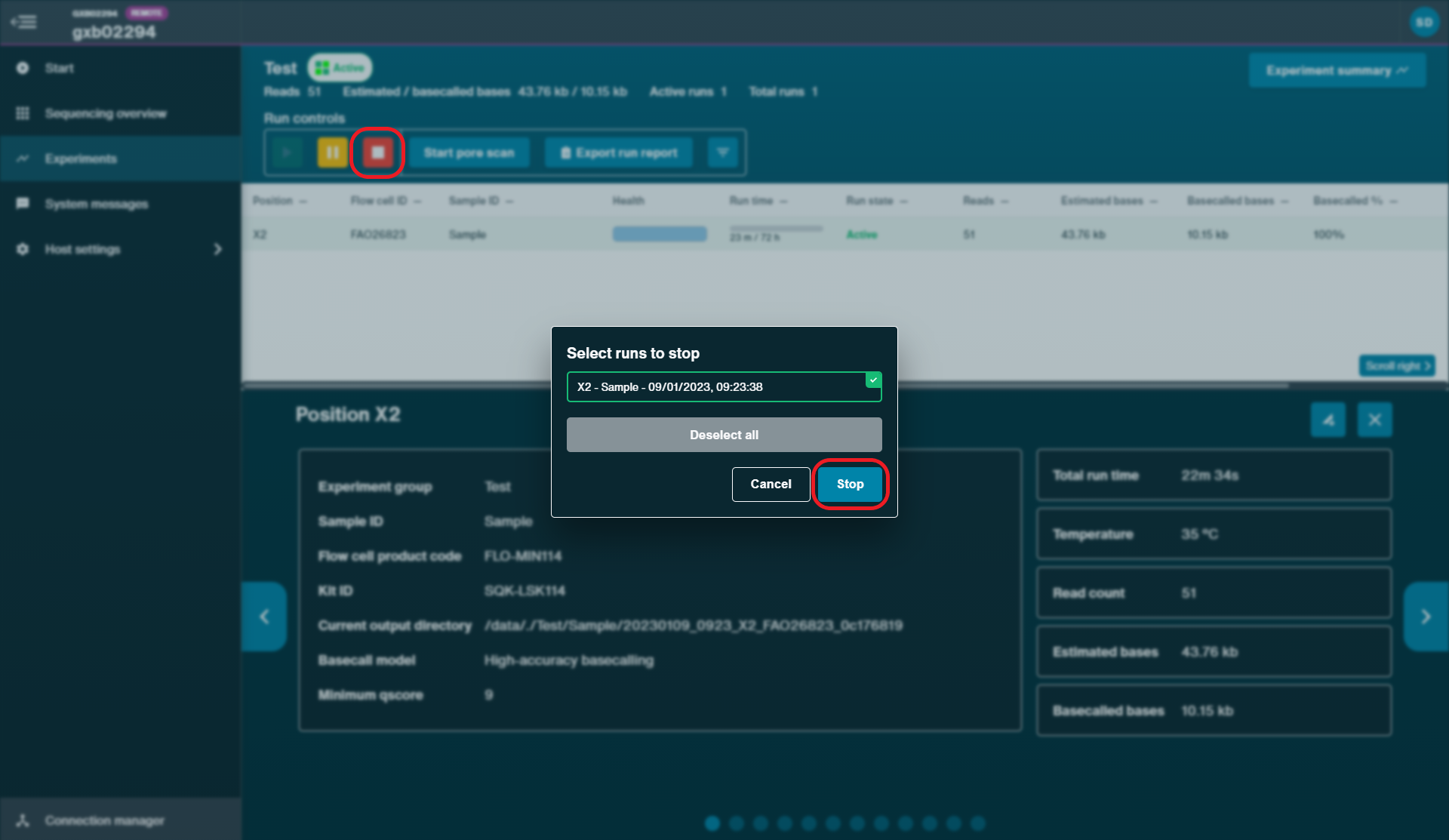
-
Thaw and prepare the flow cell priming mix according to the "Priming and loading the SpotON flow cell" section of the suitable protocol.
-
Open the MinION or GridION device lid and slide the second flow cell under the clip. Press down firmly on the flow cell to ensure correct thermal and electrical contact.
Note: We recommend leaving the original flow cell in the device and inserting the second flow cell in a free position, where possible.
If a free position is not available on the device:
1. Remove the original flow cell and place in the tray the flow cell was delivered in. Ensure the flow cell remains as level as possible to prevent waste fluid leaking out of the waste port.
2. Insert the second flow cell in the device for priming.
3. Library recovery can be completed from the original flow cell in the plastic tray. -
To prime the second flow cell, slide the priming port cover clockwise to open the priming port.
-
After opening the priming port, check for a small air bubble under the cover. Draw back a small volume to remove any bubbles:
- Set a P1000 pipette to 200 µl
- Insert the tip into the priming port
- Turn the wheel until the dial shows 220-230 µl, to draw back 20-30 µl, or until you can see a small volume of buffer entering the pipette tip
Note: Visually check that there is continuous buffer from the priming port across the sensor array.
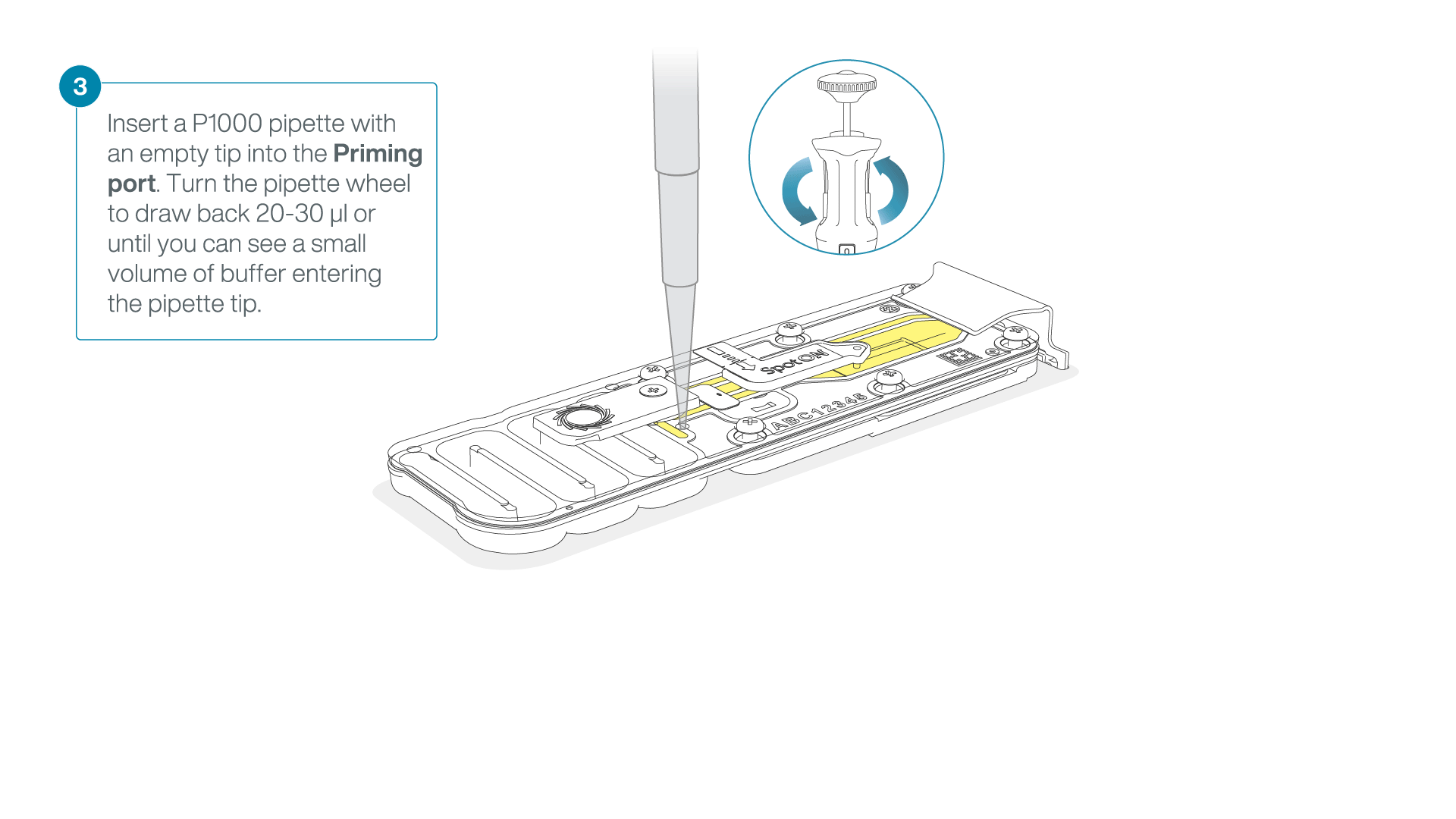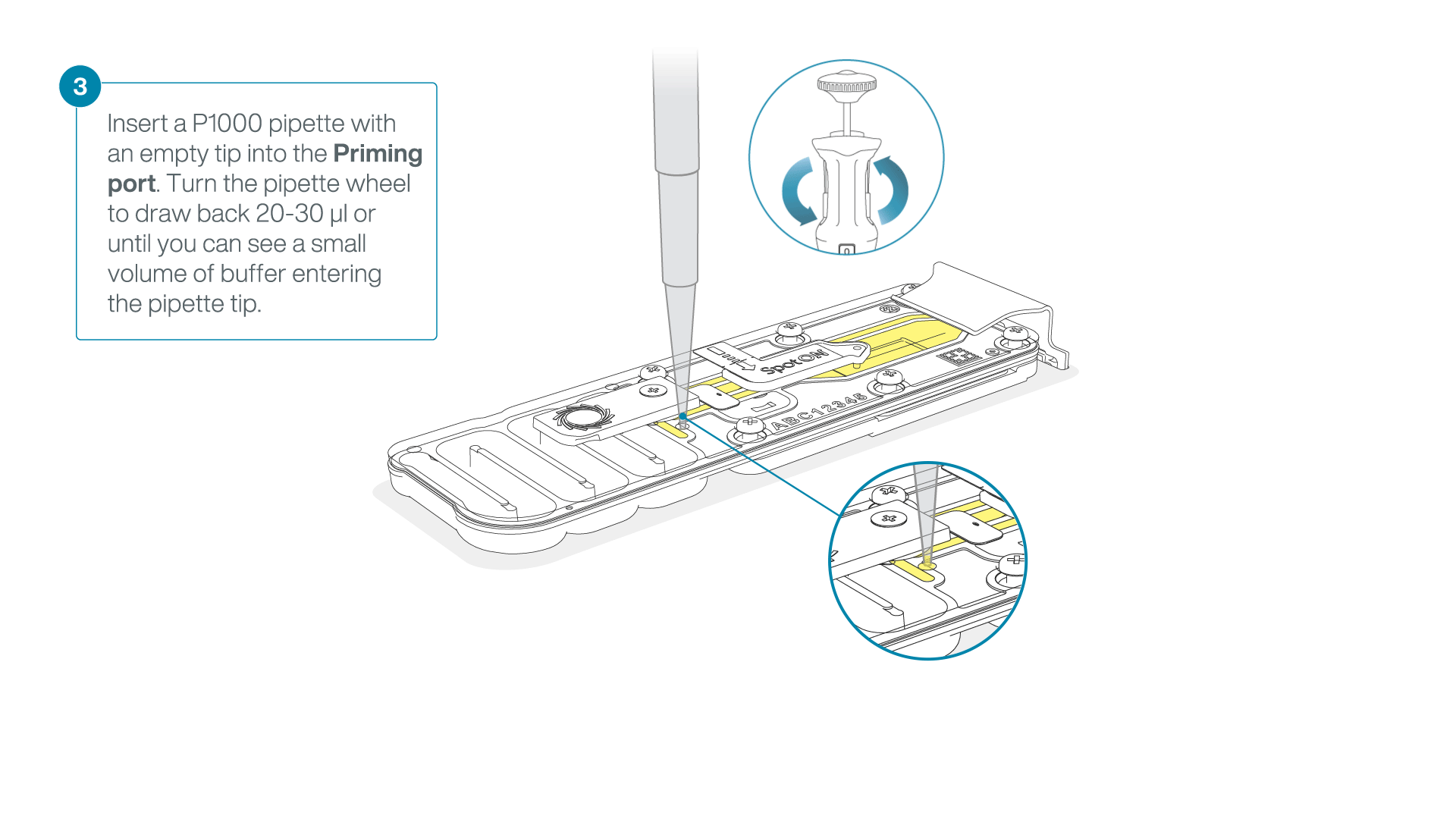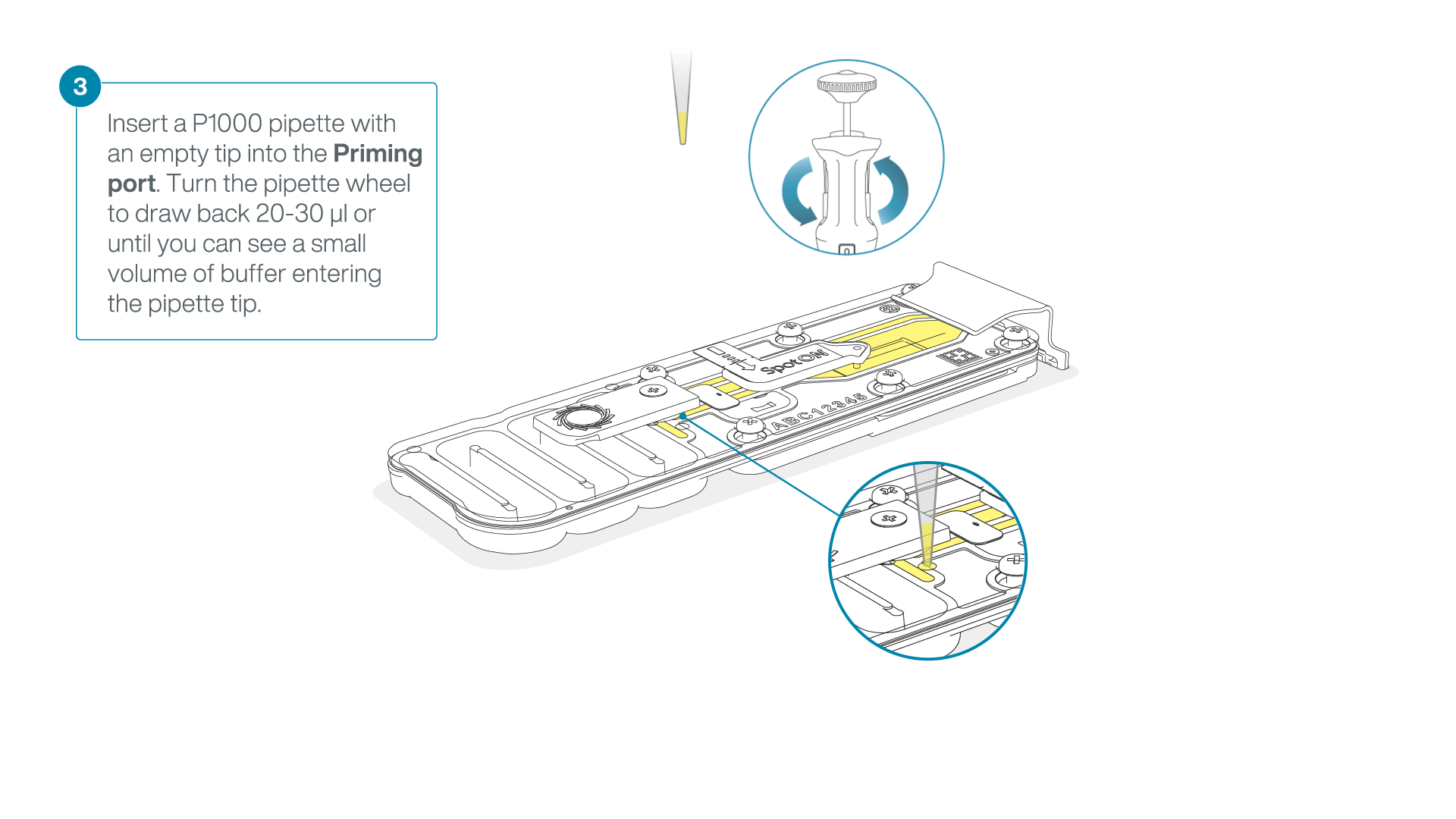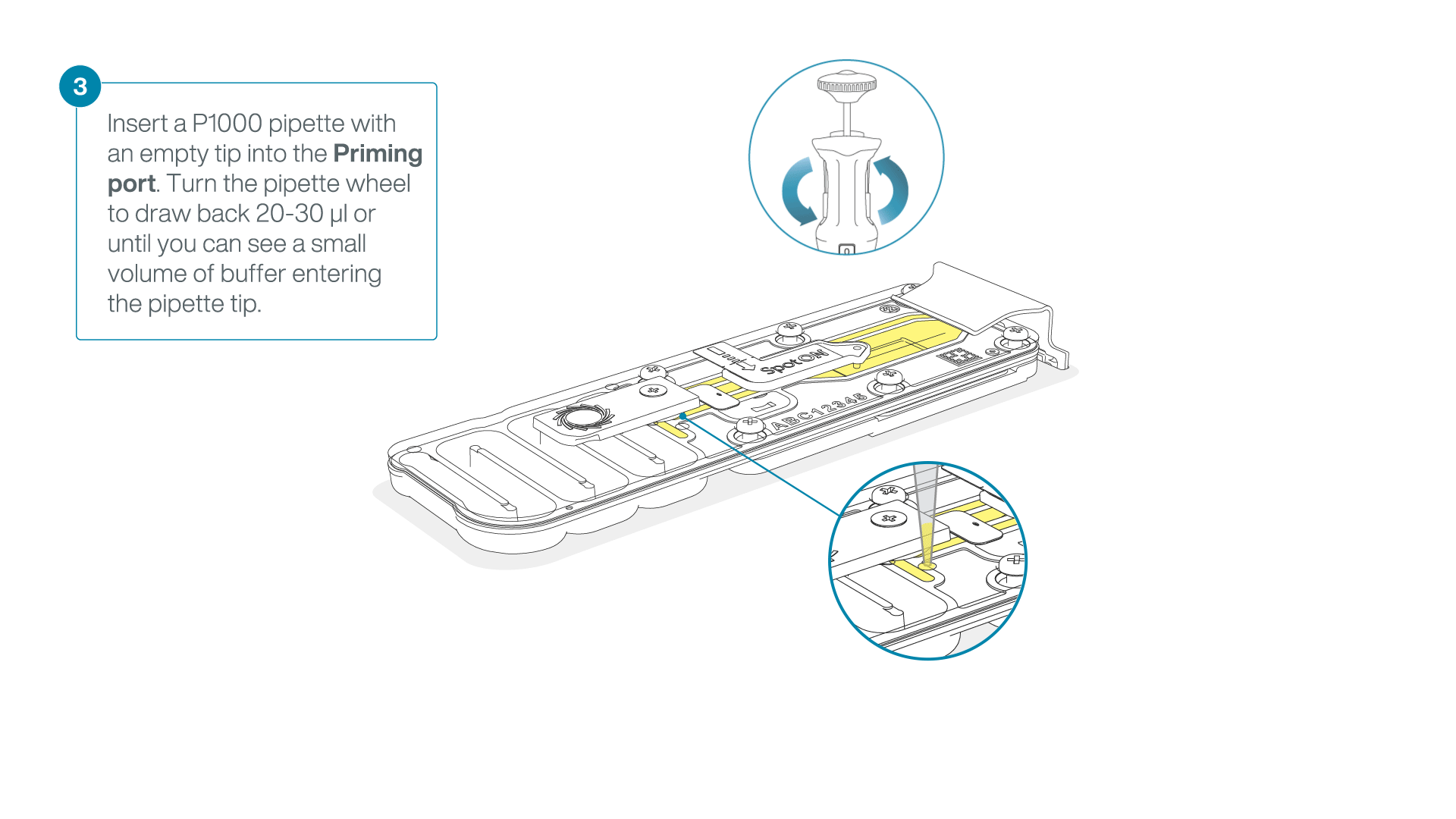
-
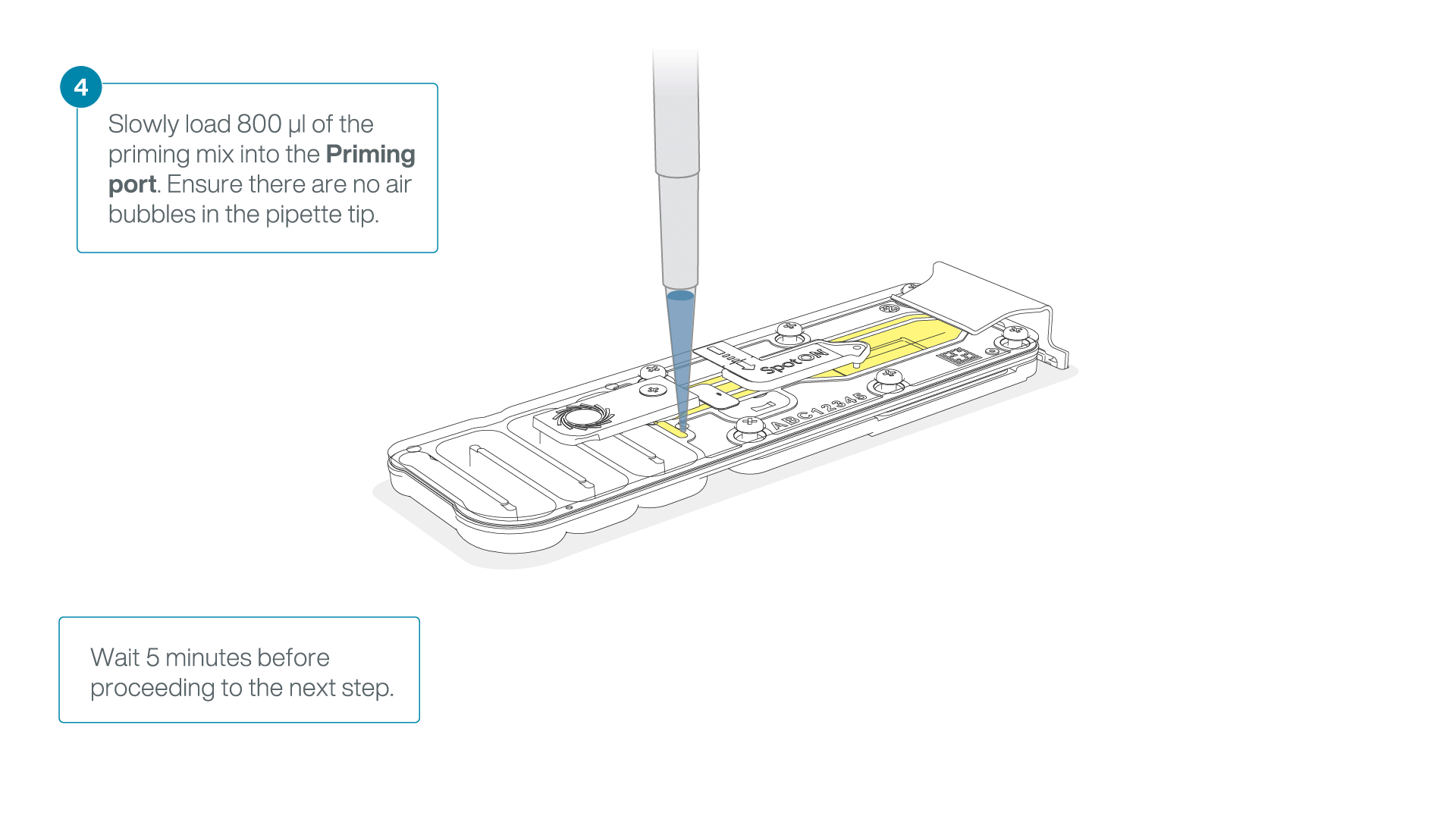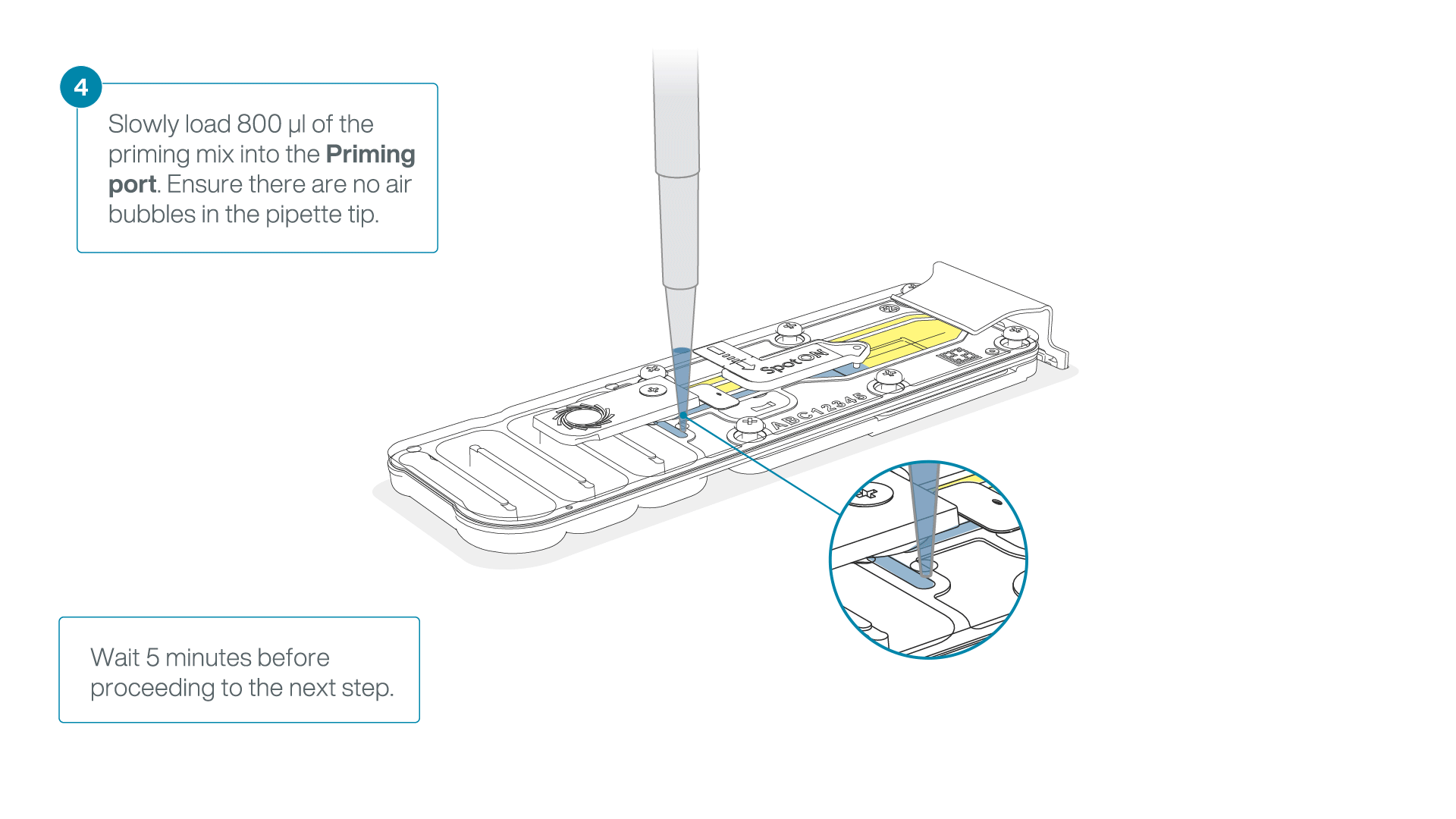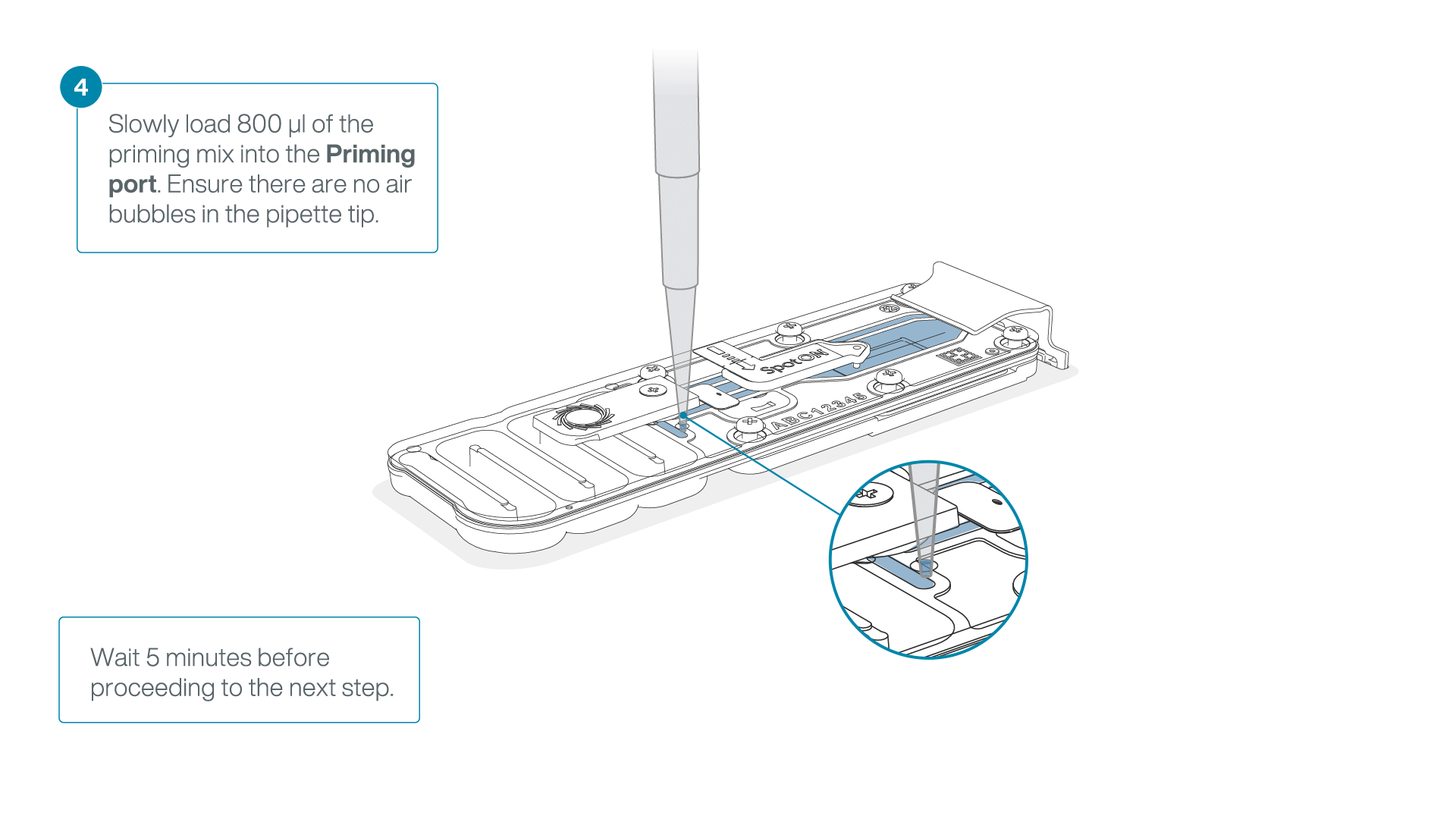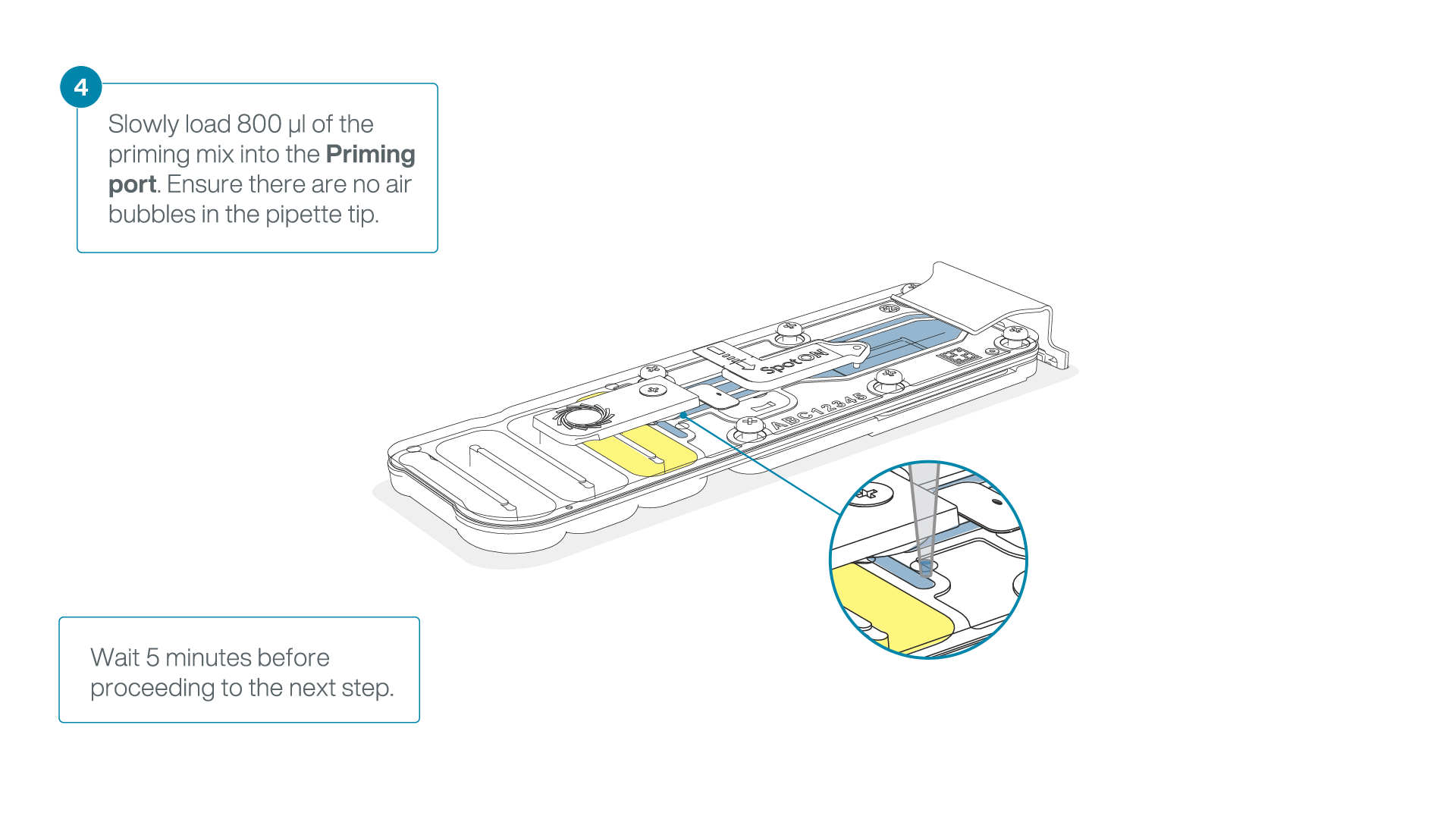
Load 800 µl of the priming mix into the second flow cell via the priming port, avoiding the introduction of air bubbles. Wait for five minutes.
-
Complete the flow cell priming for the second flow cell:
- Gently lift the SpotON sample port cover to make the SpotON sample port accessible.
- Load 200 µl of the priming mix into the flow cell via the priming port (not the SpotON sample port), avoiding the introduction of air bubbles.
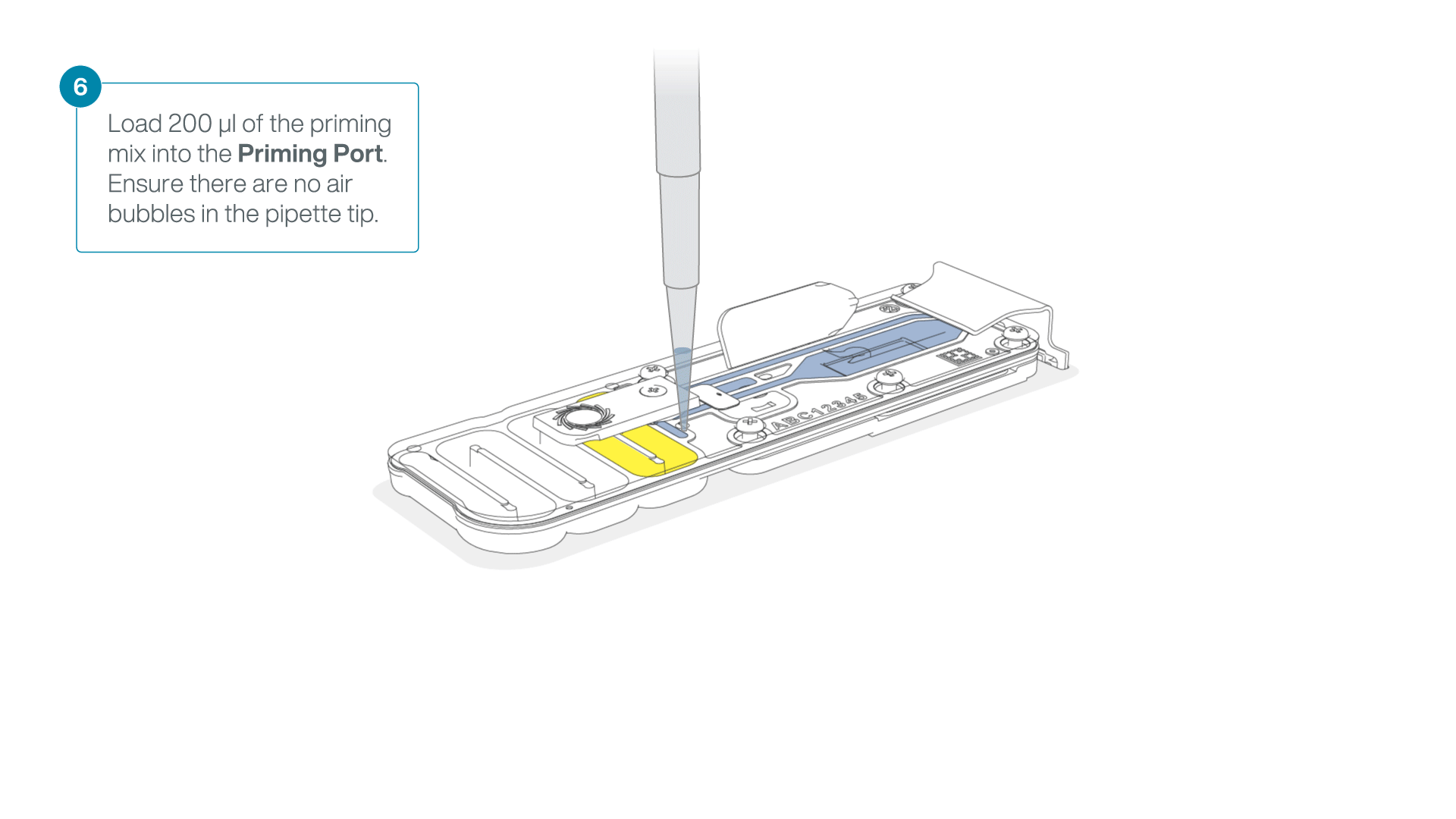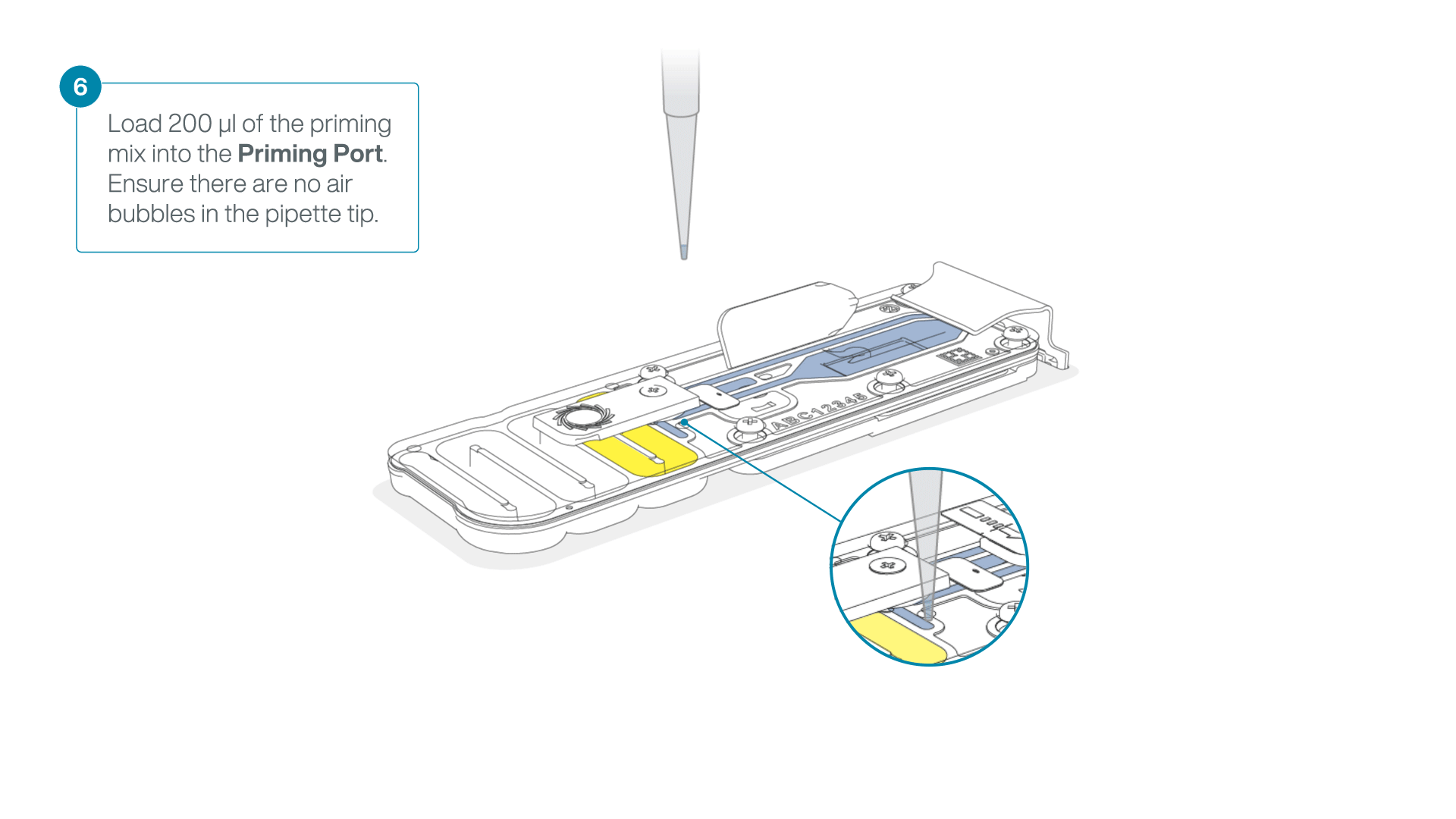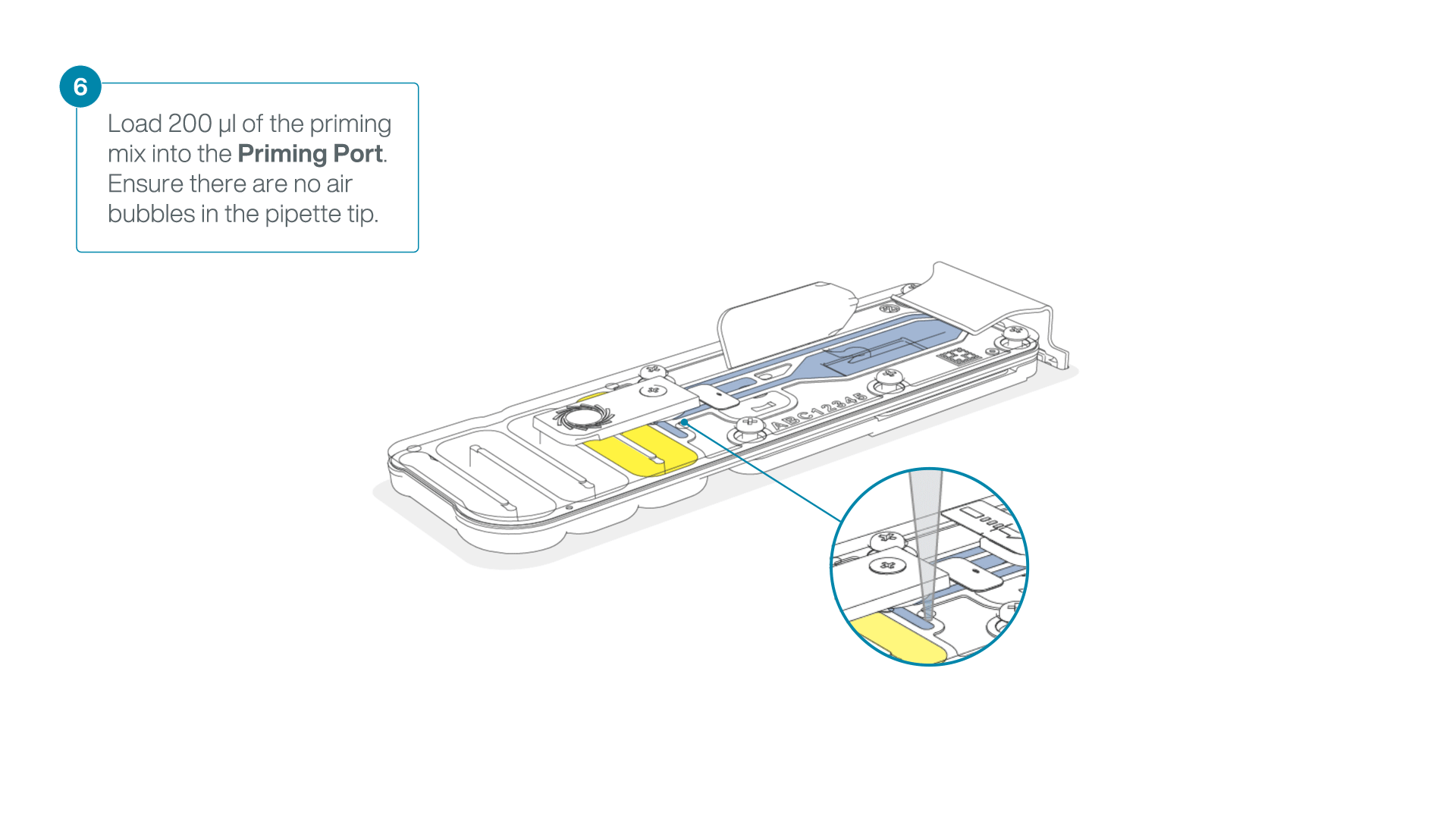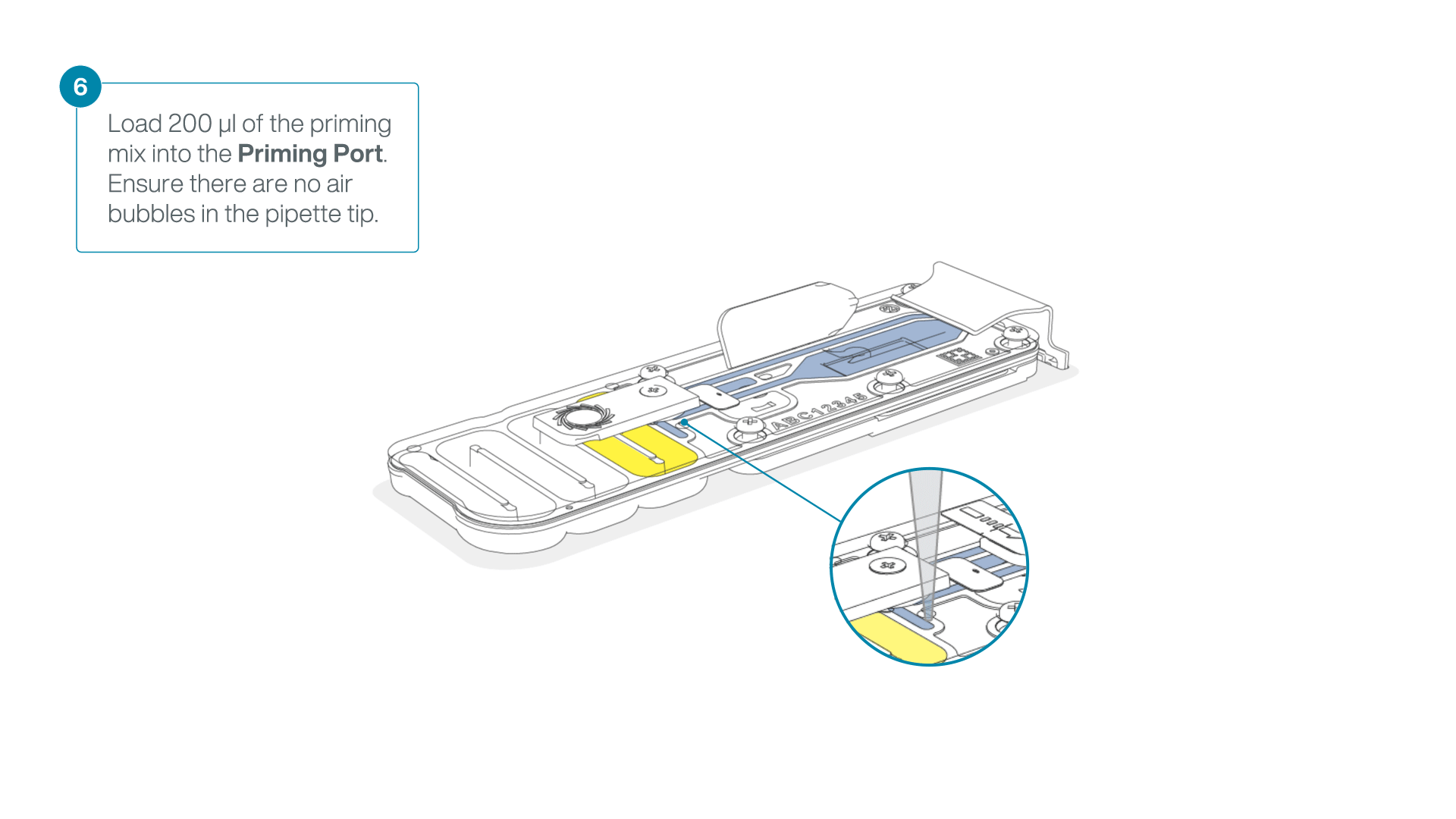
-
To prepare the original flow cell for library recovery, slide open the priming port cover and lift open the SpotON sample port cover.
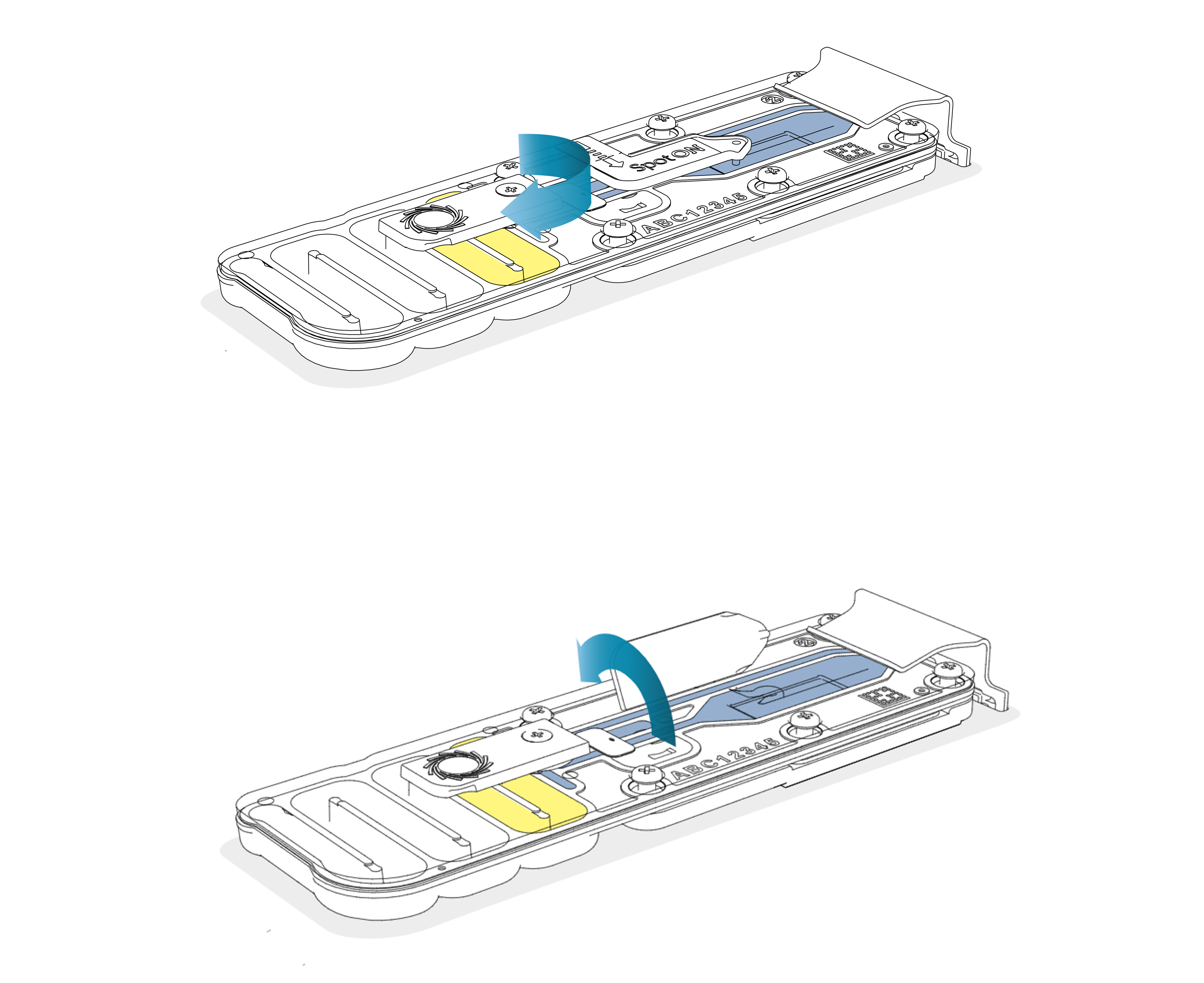
-
Set a pipette to 75 µl and fully depress the pipette before inserting the tip into the SpotON port of the original flow cell. Slowly aspirate to recover the DNA library from the flow cell.
Note: Insert the pipette tip to the point where it is touching the liquid in the flow cell. Do not insert the tip too far into the port as this will impact removal.
The recovered library may appear slightly yellow and not all the library loading beads (LB, LBII, LIB) will be fully recovered but this will not impact library recovery and transfer.
-
Add the recovered DNA library to the second flow cell via the SpotON sample port in a dropwise fashion. Ensure each drop flows into the port before adding the next.
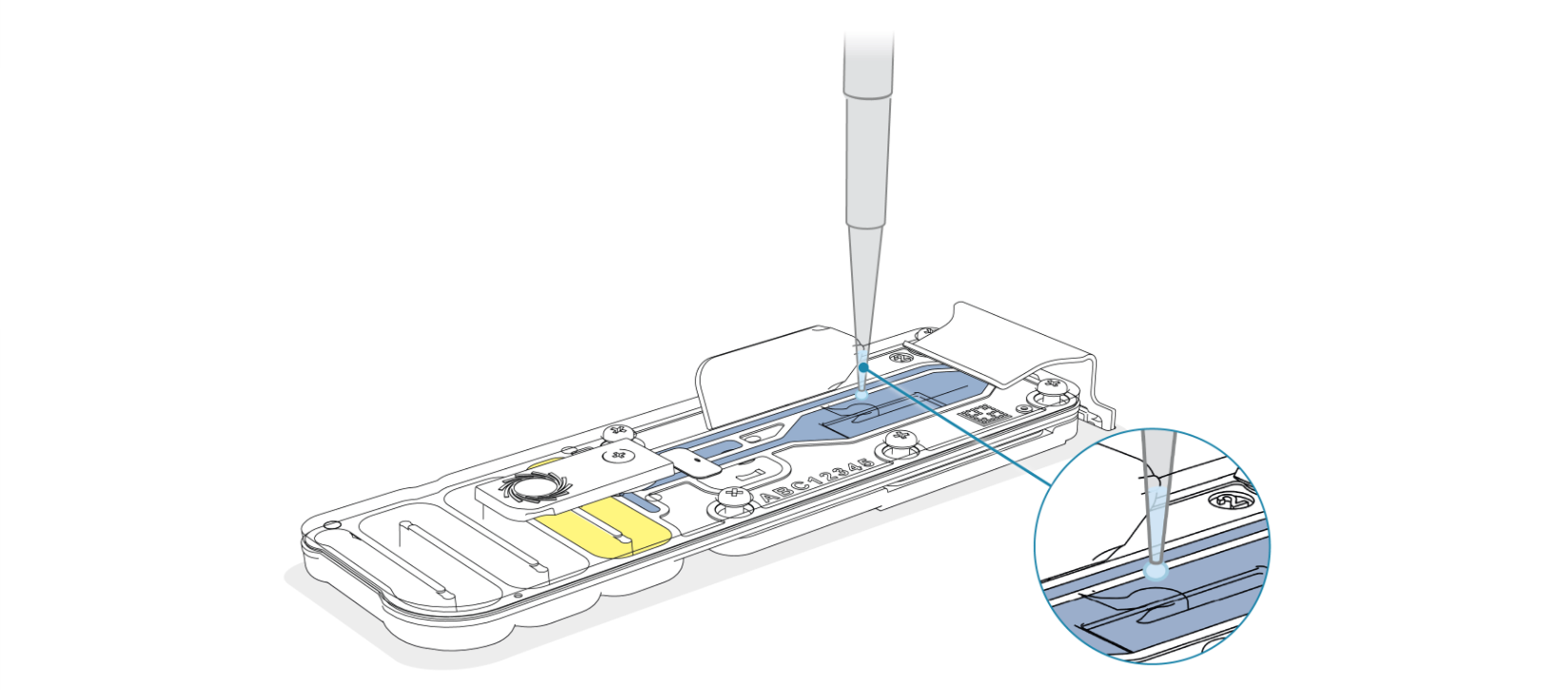
-
Gently replace the SpotON sample port cover of the second flow cell, making sure the bung enters the SpotON port and close the priming port.
-
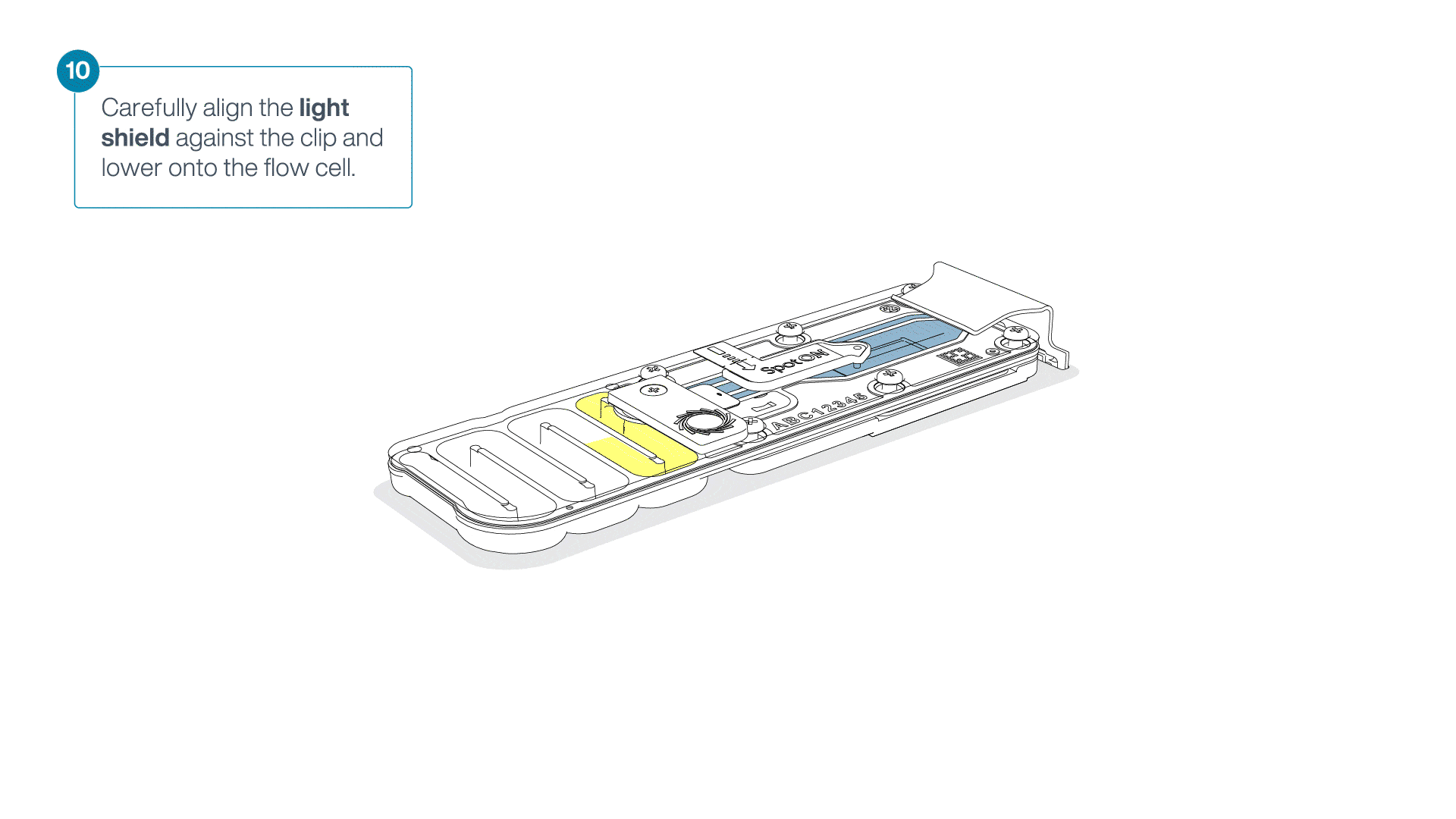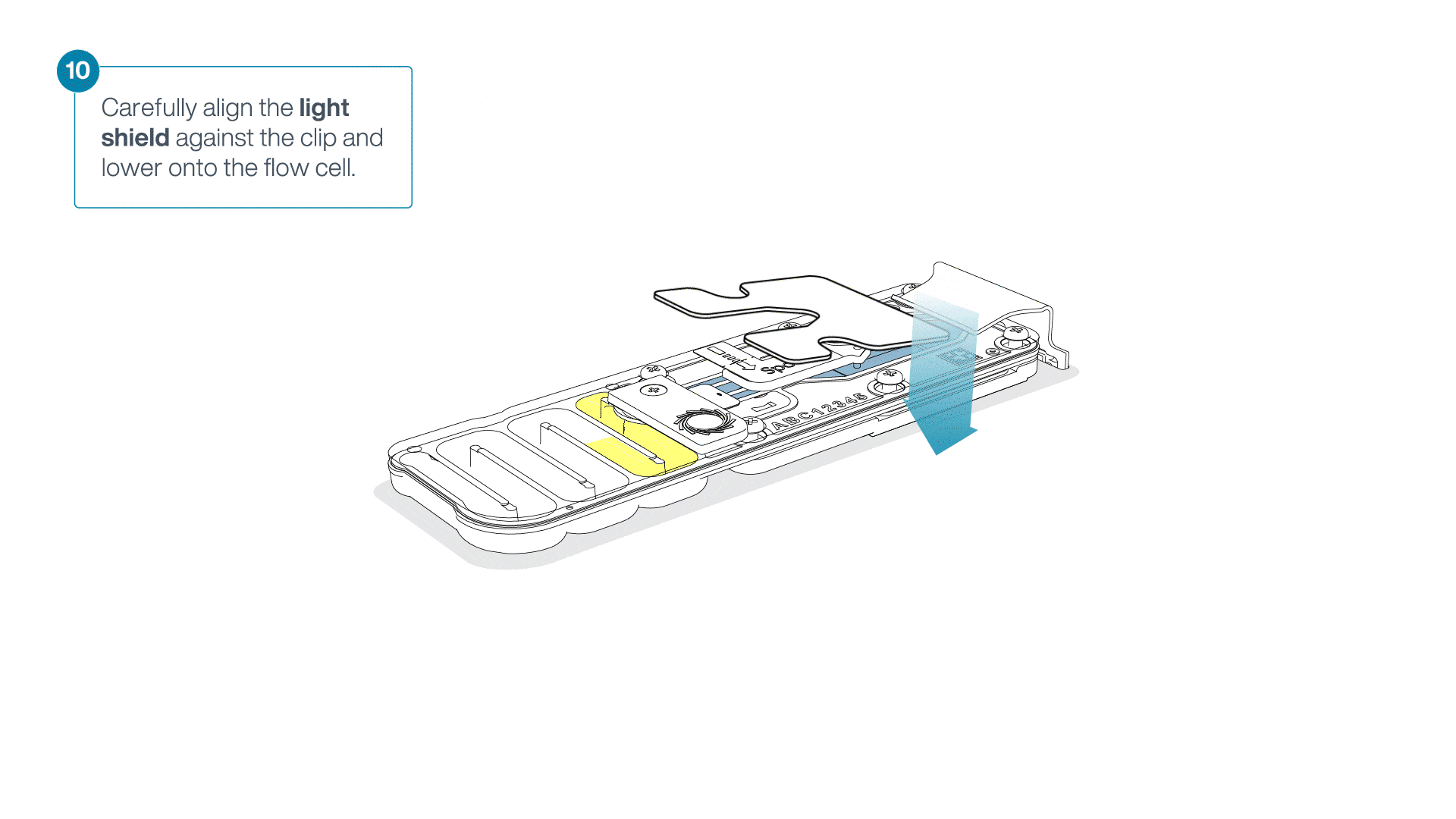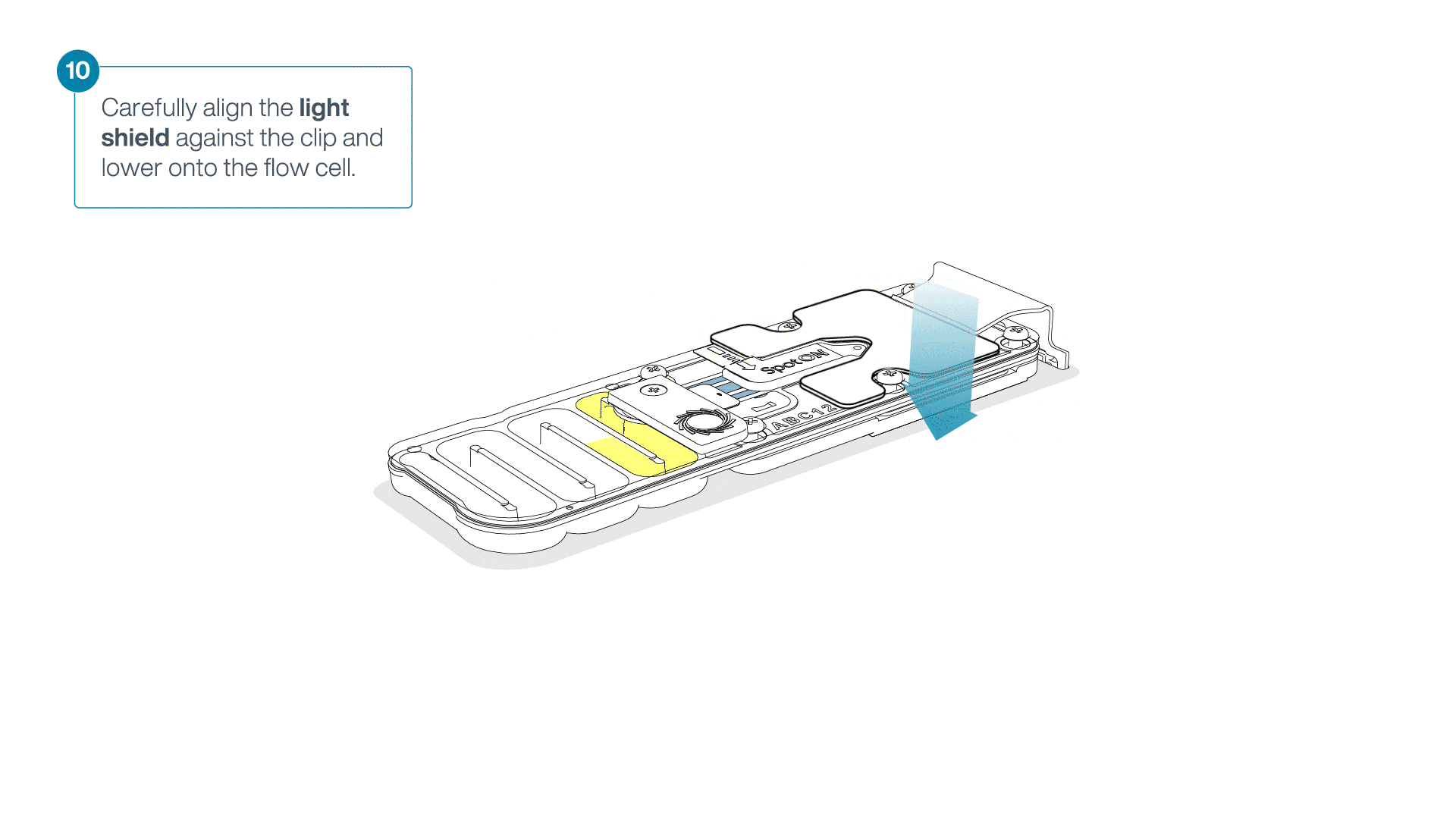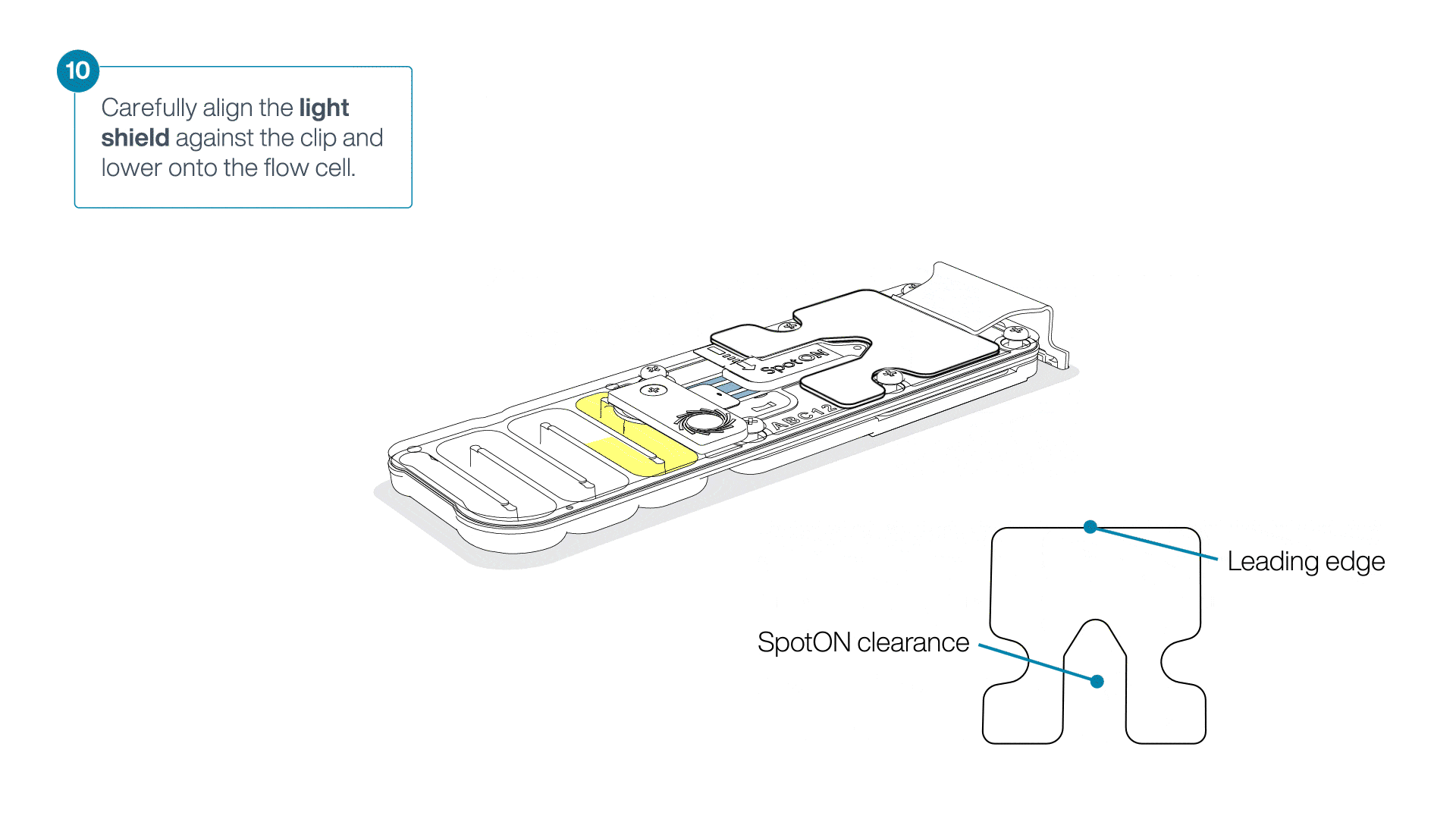
Place the light shield onto the flow cell, as follows:
Carefully place the leading edge of the light shield against the clip.
Note: Do not force the light shield underneath the clip.Gently lower the light shield onto the flow cell. The light shield should sit around the SpotON cover, covering the entire top section of the flow cell.
-
The original flow cell can be flushed with deionised water and returned to Oxford Nanopore.
Instructions for returning flow cells can be found here.
-
Start a new sequencing run on MinKNOW for the second flow cell.






