- Materials
-
- Rapid Barcode Plate (RB96)
- AMPure XP Beads (AXP, or SPRI)
- Elution Buffer from the Oxford Nanopore kit (EB)
- Rapid Adapter F (RAP F)
- Consumables
-
- Nuclease-free water (e.g. ThermoFisher, cat # AM9937)
- Freshly prepared 80% ethanol in nuclease-free water
- Bravo Lab Disposable Pipette Tips 250 µl - compatible with Bravo 96LT head (19477-022)
- Arvensis B-Frame BIOCOMPOSITE 96 Well PCR Plate Fully Skirted Low Profile 0.2 ml wells
- 96-well 0.8 ml MIDI plate (we recommend Abgene™ 96 Well 0.8 ml Polypropylene Deepwell Storage Plate: ThermoFisher, Cat # AB0859)
- PCR plate seals
- Qubit™ Assay Tubes (Invitrogen, Q32856)
- Qubit dsDNA HS Assay Kit (Invitrogen, Q32851)
- Equipment
-
- Agilent Bravo liquid handling robot
- Thermal cycler
- Centrifuge capable of taking 96-well plates
- P1000 pipette and tips
- P200 pipette and tips
- P20 pipette and tips
- P2 pipette and tips
- Qubit fluorometer (or equivalent for QC check)
-
Rapid Barcoding:
-
Select the 'Barcoding and clean-up' program on the Agilent Bravo.
-
Set the number of columns for your run:
- For three columns of each primer pool (6 in total), select 3 columns.
- For six columns of each primer pool (12 in total), select 6 columns.
- For twelve columns of each primer pool (24 in total), select 12 columns.
-
Select 'Display Deck Layout'.
-
Add the Primer plates, the Rapid barcode plate and the labware as indicated on the display.
-
Select 'Run protocol'.
-
To start the run, select 'Ok' from the figure below:
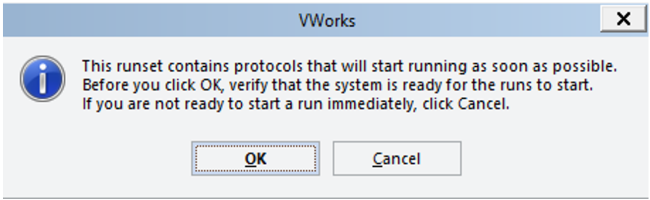
-
Optional actionFor runs using X24 or X48 samples, you will need to enter the starting index plate column for the RBK plate when prompted.
For example: If your barcode starts at column 4, enter "4" as seen in the figure below.
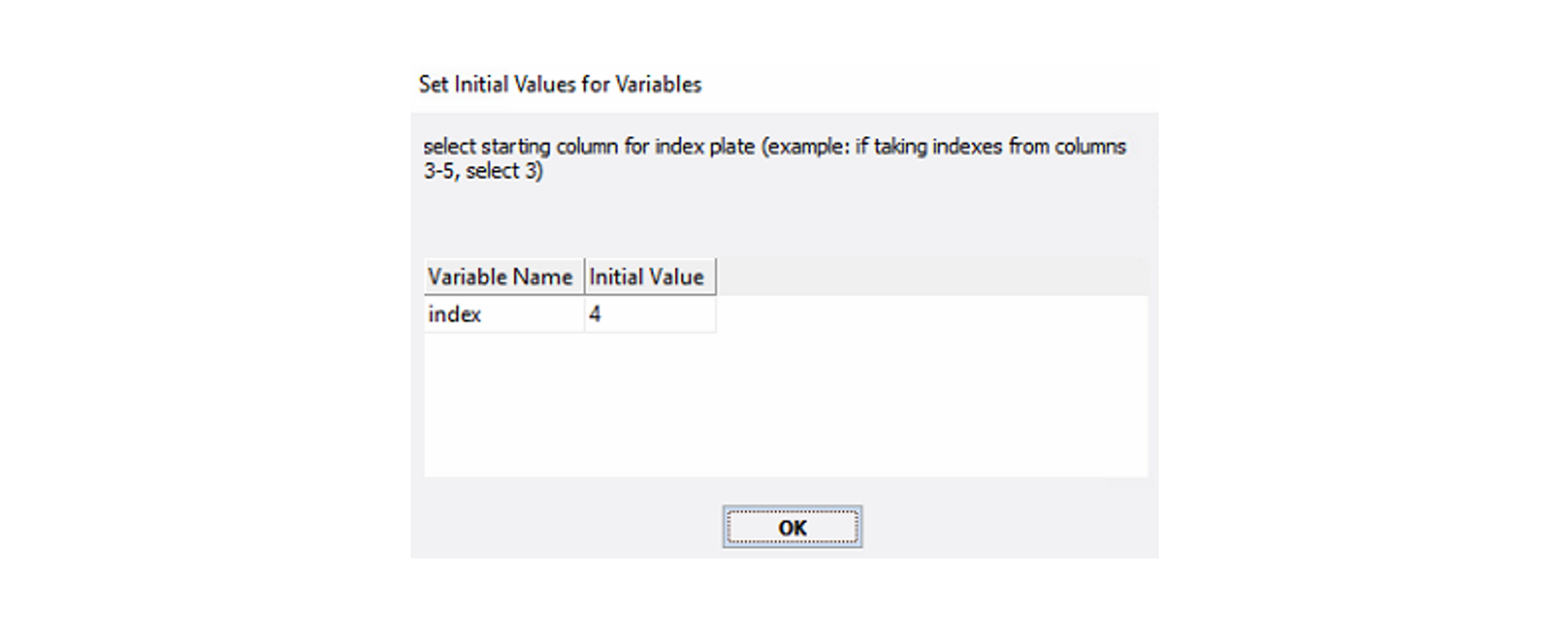
-
After the Agilent Bravo has completed the Rapid Barcoding plate, the robot will stop with the following message:
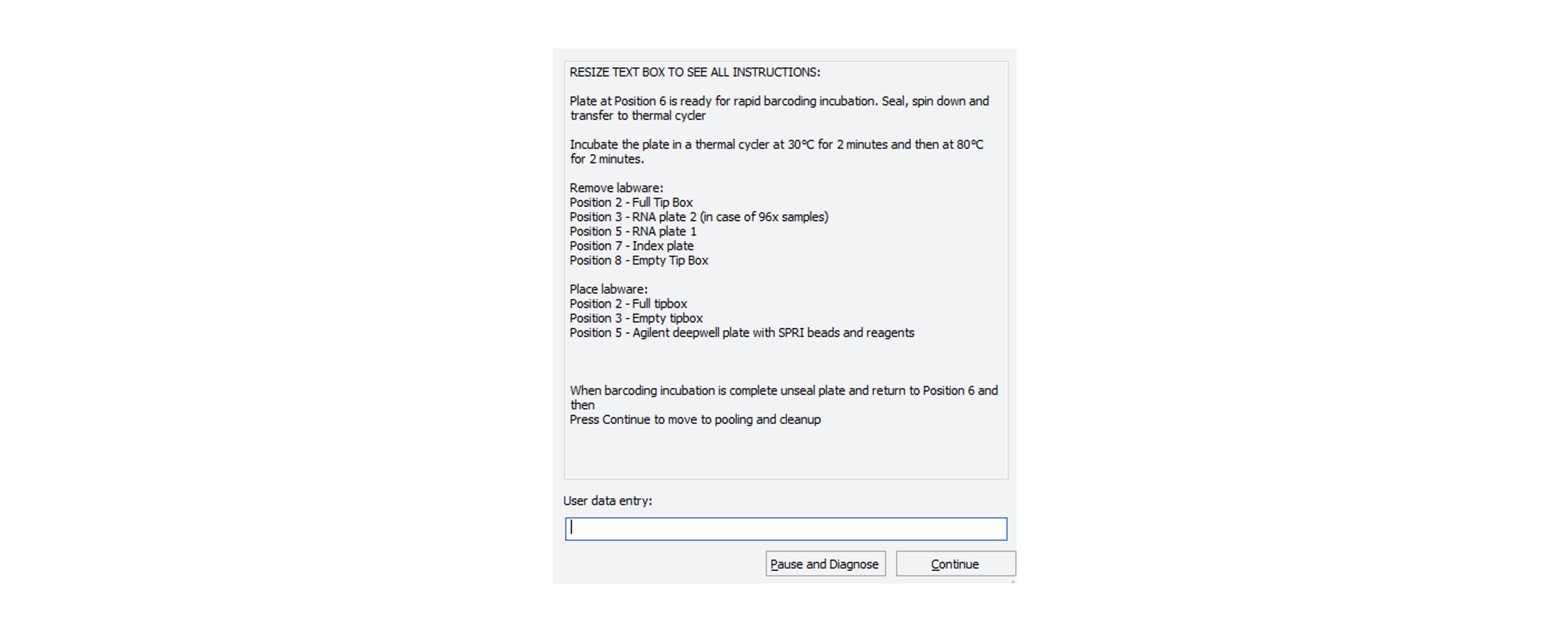
-
Remove the barcoded sample plate from the Agilent Bravo, seal it and spin it down.
-
Place the barcoded sample plate in the thermal cycler and incubate at 30°C for 2 minutes and then at 80°C for 2 minutes.
-
Sample pooling and clean-up:
-
Follow the instructions for the Agilent Bravo deck set-up on the screen prompt.

-
Resuspend the AMPure XP beads by vortexing.
-
While the barcoding plate is in the thermal cycler, using a clean MIDI 96 deep-well plate prepare the reagents as follows:
For X24 samples:
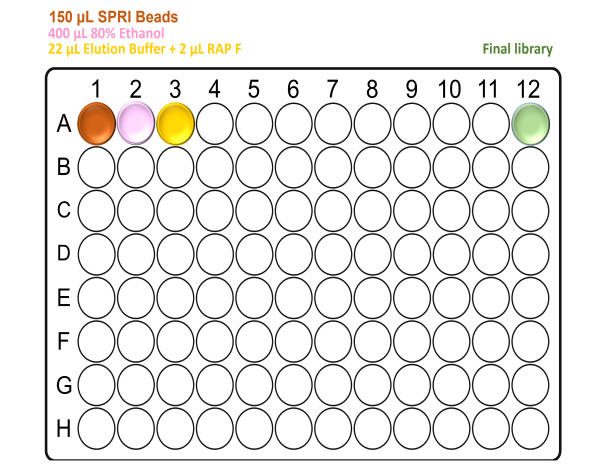
For X48 samples:
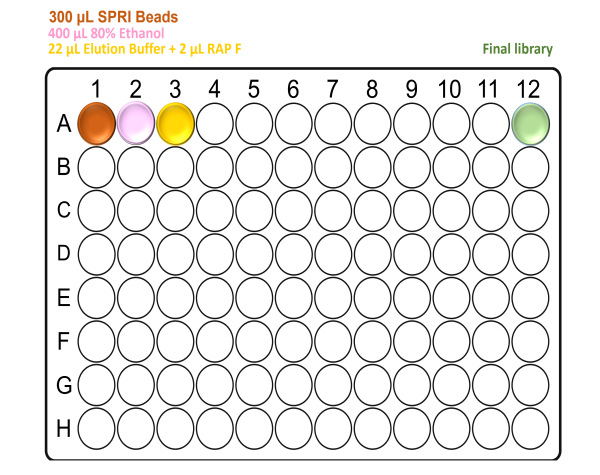
For X96 samples:
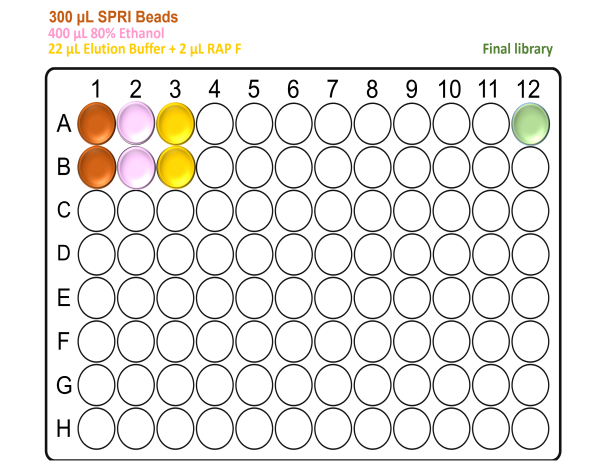
Note: Take care to not introduce air bubbles while aliquoting the reagents into the deep-well plate.
Ensure all reagents are properly mixed prior to use.
When preparing the plates, well A12 will be empty. This is where the final elution will be found at the end of the Agilent Bravo run.
-
Prepare the deck of the Agilent Bravo with the reagent plate and labware as follows:
For X24 or X48 samples:
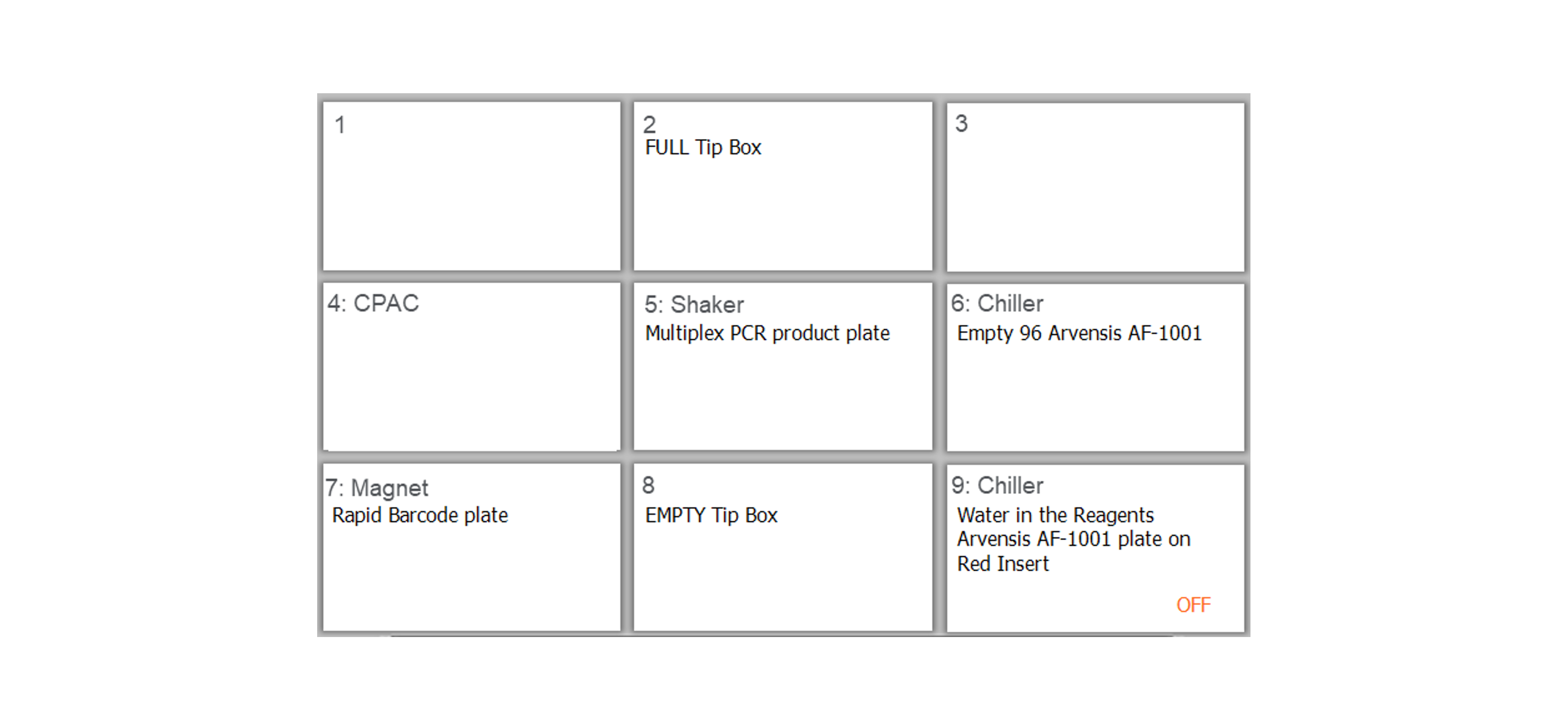
For X96 samples:

-
Once the thermal cycler has finished, remove the plate and spin it down.
-
Remove the seal on the rapid barcoding plate and return it to the allocated position on the Agilent Bravo deck.
-
Select "Continue" on the Agilent Bravo screen prompt to continue the run.
Ensure all of the labware and reagent plates are in the correct positions, you have removed all of the lids and the plates are unsealed before continuing.
-
After the run ends, the final library can be collected from position A12 in the deep-well plate. Remove and retain the final elution into a clean 1.5 ml Eppendorf DNA LoBind tube
-
Quantify DNA concentration of the final elution by using the Qubit dsDNA HS Assay Kit.







