- Materials
-
- Input RNA in 10 mM Tris-HCl, pH 8.0
- Midnight Primer Pool A (MP A)
- Midnight Primer Pool B (MP B)
- Consumables
-
- Arvensis B-Frame BIOCOMPOSITE 96 Well PCR Plate Fully Skirted Low Profile 0.2 ml wells
- Bravo Lab Disposable Pipette Tips 250 µl - compatible with Bravo 96LT head (19477-022)
- PCR plate seals
- Nuclease-free water (e.g. ThermoFisher, cat # AM9937)
- LunaScript RT SuperMix (LS RT)
- 1.5 ml Eppendorf DNA LoBind tubes
- Q5 HS Master Mix (Q5)
- Equipment
-
- Ice bucket with ice
- PCR hood with UV steriliser (optional but recommended to reduce cross-contamination)
- Agilent Bravo liquid handling robot
- P1000 pipette and tips
- P200 pipette and tips
- P20 pipette and tips
- P2 pipette and tips
- Microfuge
- Thermal cycler
- Centrifuge capable of taking 96-well plates
-
Run setup:
-
Turn on the Agilent Thermocube and set Position 4 to 4°C.
To cool position 4 prior to the run: open the file, under the deck layout, and tick the box near the block you want to cool.
Set up the temperature and hit 'Run' to execute.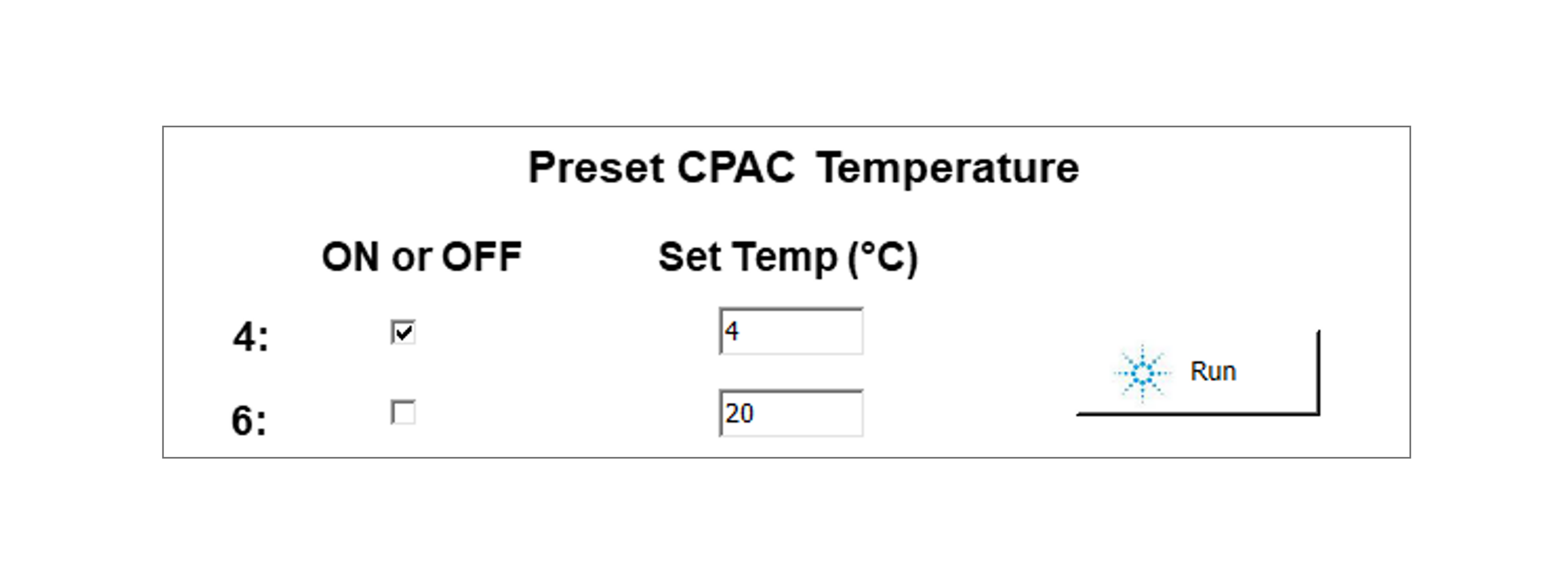
-
Thaw and keep the samples, LunaScript, Q5 Hot Start Master mix, Midnight Primer Pool A (MPA) and Midnight Primer Pool B (MPB) on ice.
-
In the template-free pre-PCR hood, prepare the following Primer master mixes in 1.5 ml Eppendorf DNA LoBind tubes and mix thoroughly as follows:
For x24 samples:
Reagent Pool A Pool B Nuclease-free water 172 µl 172 µl Midnight Primer Pool A (MP A) 2 µl - Midnight Primer Pool B (MP B) - 2 µl Q5 HS Master Mix (Q5) 102 µl 102 µl Total 276 µl 276 µl For x48 samples:
Reagent Pool A Pool B Nuclease-free water 344 µl 344 µl Midnight Primer Pool A (MP A) 3 µl - Midnight Primer Pool B (MP B) - 3 µl Q5 HS Master Mix (Q5) 203 µl 203 µl Total 550 µl 550 µl For x96 samples:
Reagent Pool A Pool B Nuclease-free water 687 µl 687 µl Midnight Primer Pool A (MP A) 6 µl - Midnight Primer Pool B (MP B) - 6 µl Q5 HS Master Mix (Q5) 407 µl 407 µl Total 1,100 µl 1,100 µl Note: Taking care not to introduce air bubbles, pipette mix 10-15 times between each addition and perform a final full-volume pipette mix 10 times.
Keep on ice until use.
-
In the template-free pre-PCR hood and using a clean Arvensis plate, prepare the reagent input plate as follows:
For x24 samples:
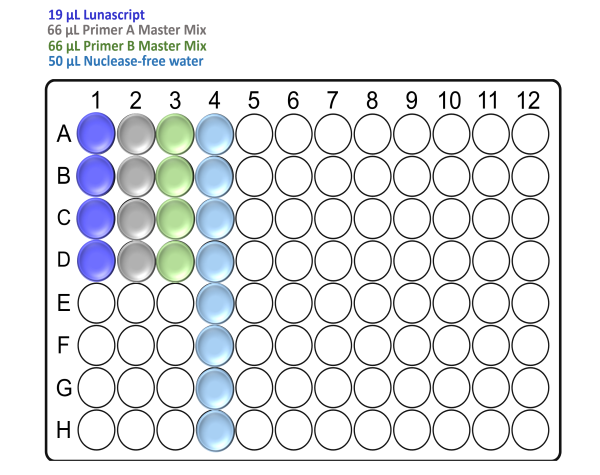
For x48 samples:
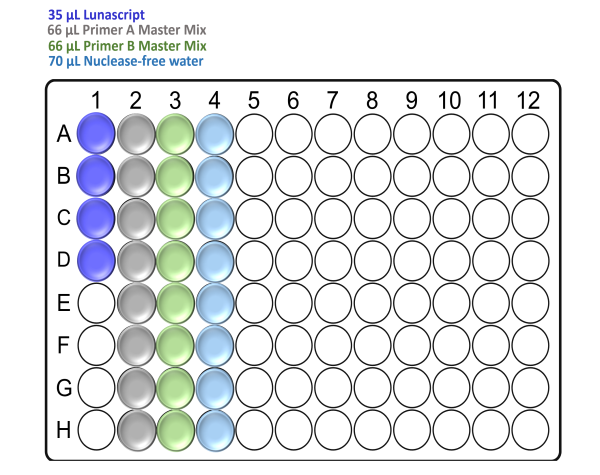
For x96 samples:
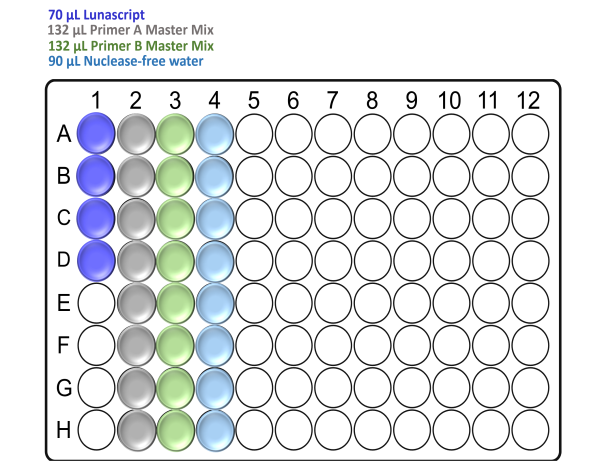
Note: Take care to not introduce air bubbles while aliquoting the reagents into the Arvensis plate.
Once complete seal the plate and keep on ice until ready to transfer over to the Agilent Bravo robot.
-
In a pre-PCR hood and using a clean Arvensis plate, prepare the RNA sample input plate as follows:
For x24 samples:
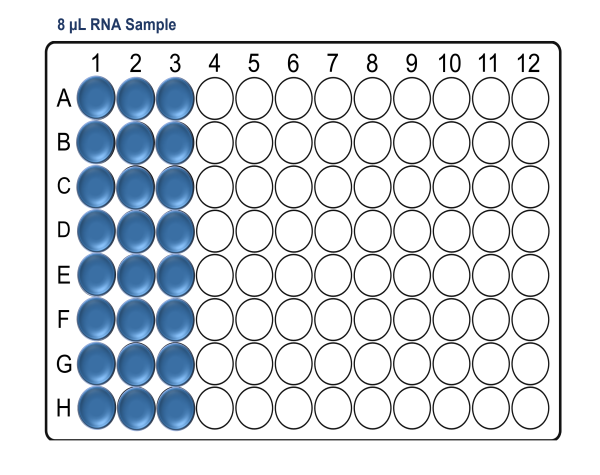
For x48 samples:
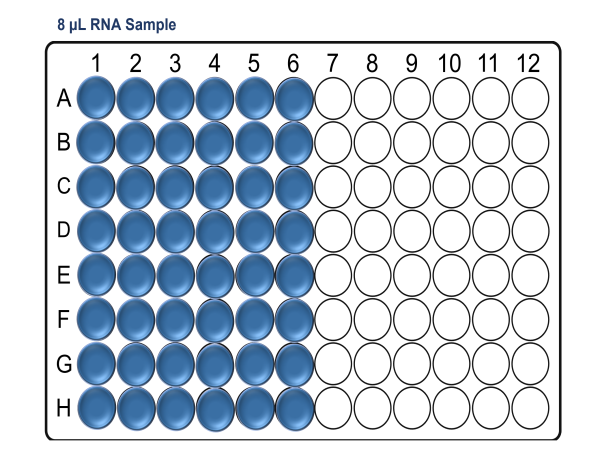
For x96 samples:
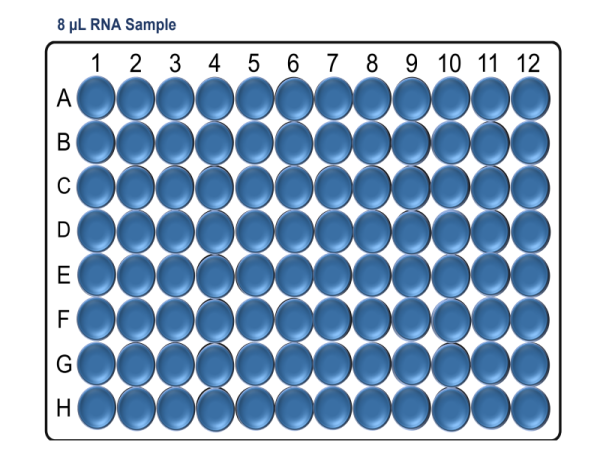
Note: Take care to not introduce air bubbles while aliquoting the samples into the Arvensis plate.
Once complete seal the plate and keep on ice until ready to transfer over to the Agilent Bravo robot.
-
On the Agilent Bravo, select the 'cDNA and Multiplex (X) samples' protocol, where (X) indicates the number of samples to be processed.
-
Set the number of columns of samples and the PCR plate type.
- For X24 samples, select 3
- For X48 samples, select 6
- For X96 samples, select 12
-
Select 'Display Deck Layout'.
-
Add the labware, sample plate and reagent input plate as indicated on the form display.
For X24 samples:
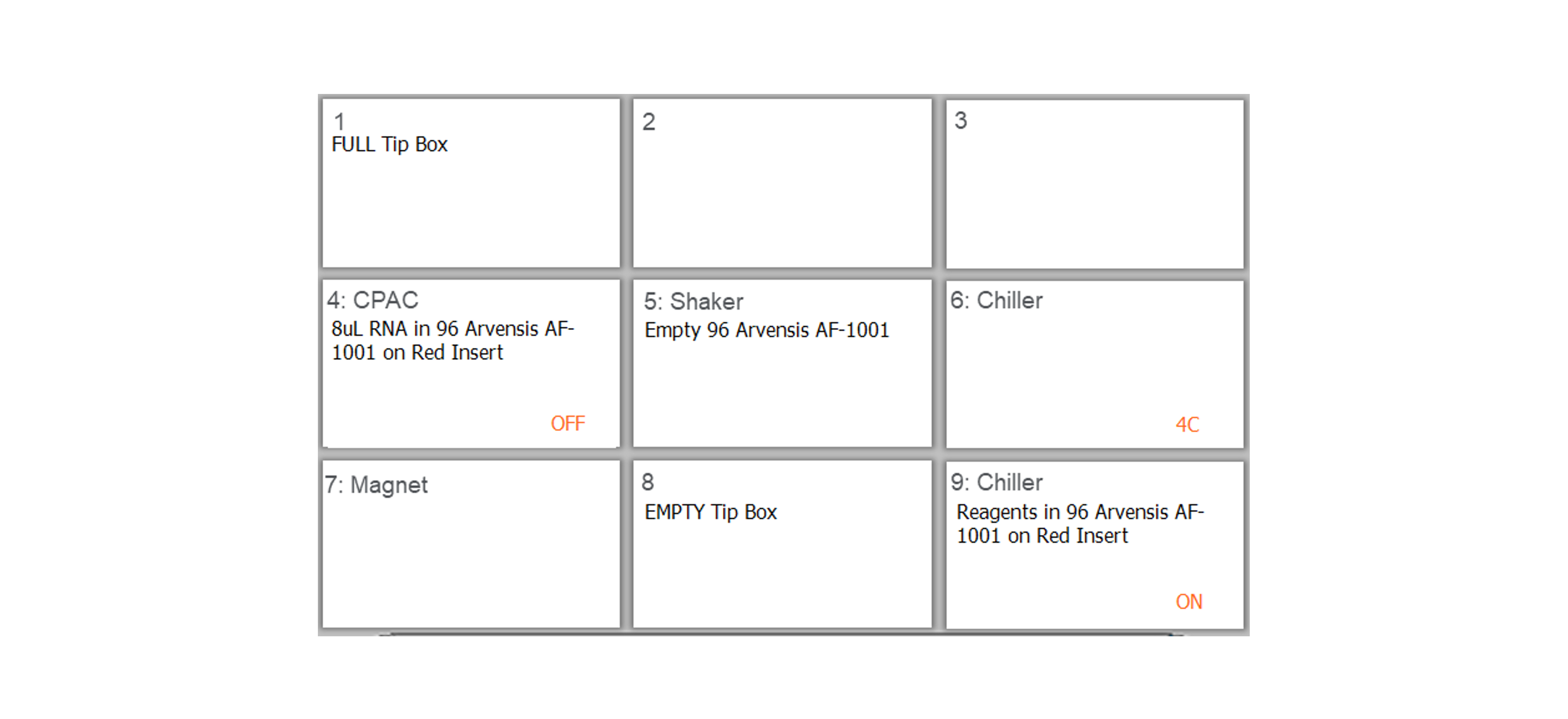
For X48 samples:

For X96 samples:

-
Select 'Run Protocol'.
-
To start the run, select 'Ok' from the figure below:
-
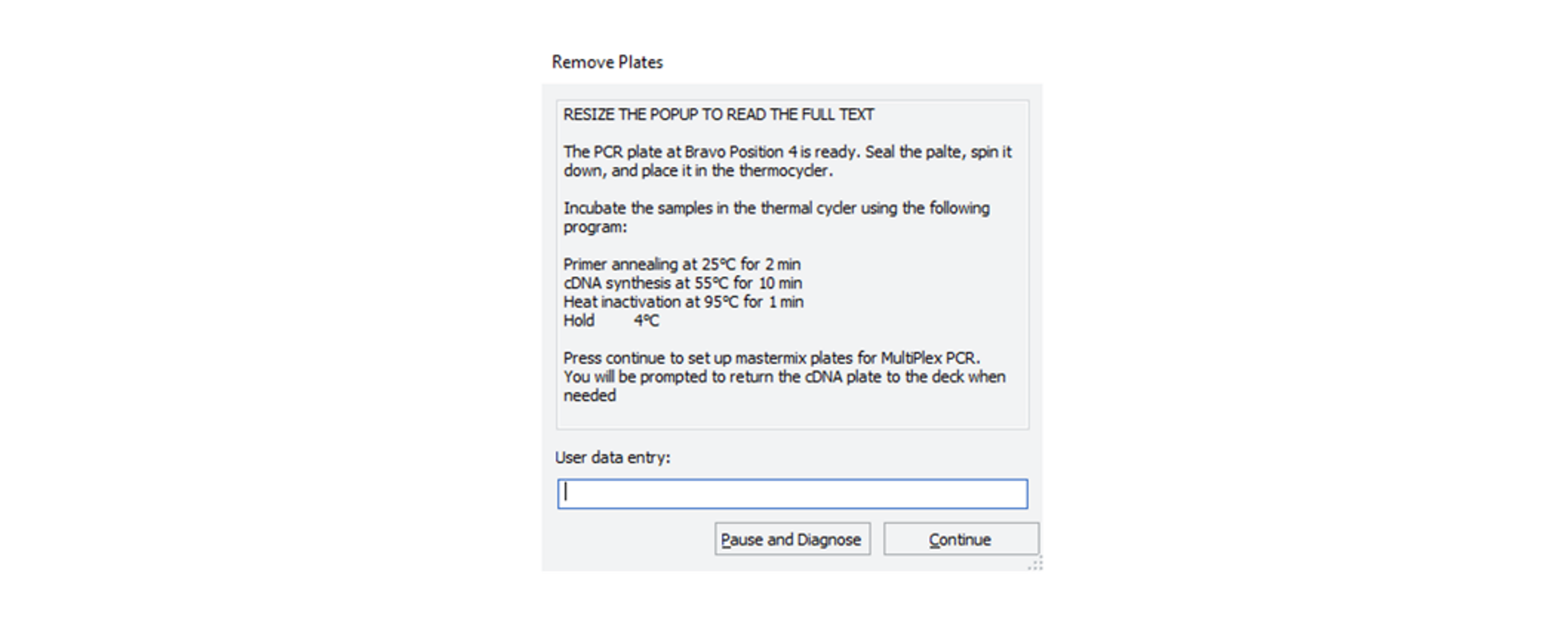
After the Agilent Bravo has added the 2 µl of LunaScript to the samples, the robot will stop with the the following message:
-
Remove the sample plate containing the LunaScript from the Agilent Bravo, seal it and spin it down.
-
Place the sample plate in a Thermal cycler and incubate using the following program:
Step Temperature Time Cycles Primer annealing 25°C 2 min 1 cDNA synthesis 55°C 10 min 1 Heat inactivation 95°C 1 min 1 Hold 4°C ∞ -
After placing the sample plate in the Thermal cycler, press "Continue" on the Agilent Bravo to start the addition of the Primer master mixes into a clean Arvensis plate.
-
Once the thermal cycler has completed the cDNA synthesis, remove the sample plate and spin down.
-
When prompted, place the sample plate back in the Agilent Bravo deck into position 4.
-
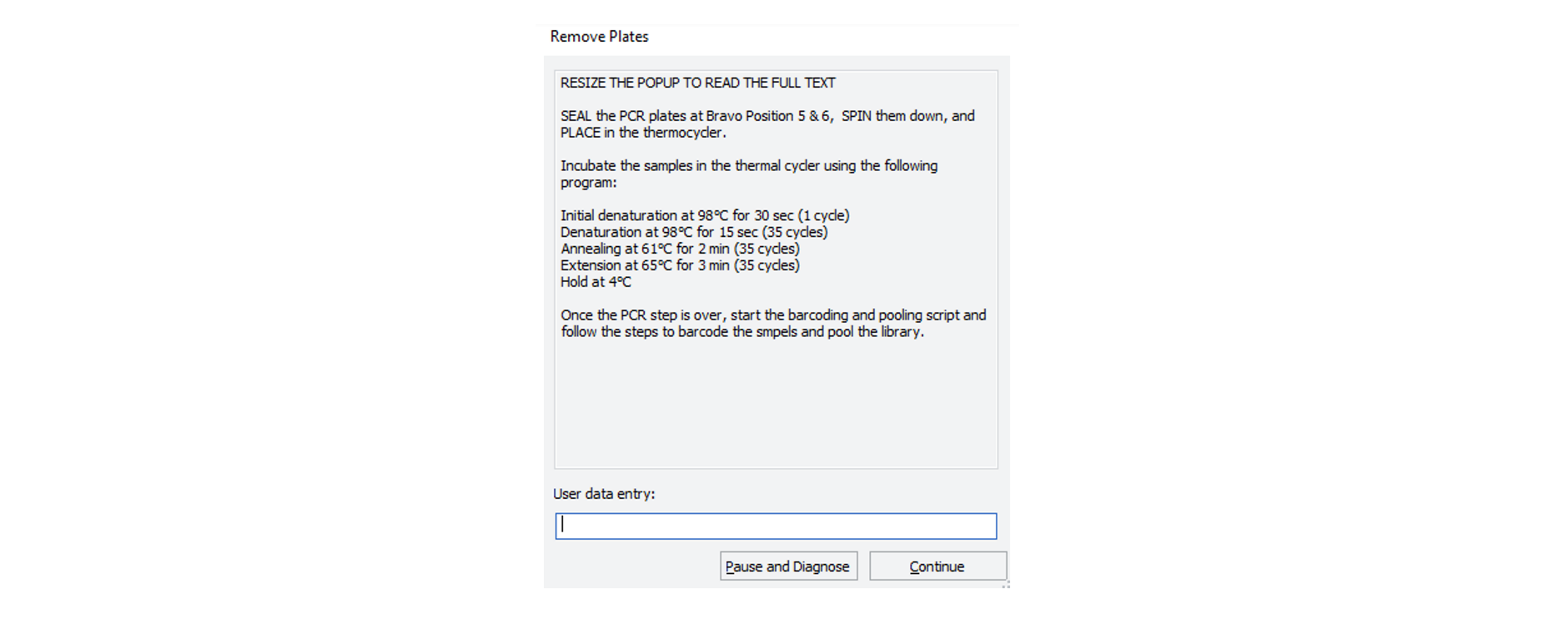
After the Agilent Bravo has added the 2.5 µl of RT to the Primer master mix plate/plates, the robot will stop with the the following message:
-
Remove the PCR plate (or plates if processing X96 samples) from the Agilent Bravo, seal it and spin it down.
-
Place in the thermal cycler and incubate using the following program, with the heated lid set to 105°C:
Step Temperature Time Cycles Initial denaturation 98°C 30 sec 1 Denaturation
Annealing and extension98°C
61°C
65°C15 sec
2 min
3 min
35Hold 4°C ∞












