- Materials
-
- Flow Cell Wash Kit XL (EXP-WSH004-XL)
- Consumables
-
- 1.5 ml Eppendorf DNA LoBind tubes
- 2 ml Eppendorf DNA LoBind tubes
- Equipment
-
- P1000 pipette and tips
- P200 pipette and tips
- P20 pipette and tips
- Ice bucket with ice
-
Preparation to run the washing procedure
An accumulation of pores in the 'recovering' state can occur ('unavailable' in the figure below). This reduces the rate of data acquisition due to fewer pores being available to accept and sequence strands. These pores can be reverted back to the 'active pore' state by pausing sequencing and washing the flow cell with the Flow Cell Wash Kit XL (EXP-WSH004-XL) (Figure 1). The wash step is only recommended when channels are lost to the 'recovering/unavailable' state.
Further information is available in the Flow Cell Wash Kit (EXP-WSH004 or EXP-WSH004-XL) protocol.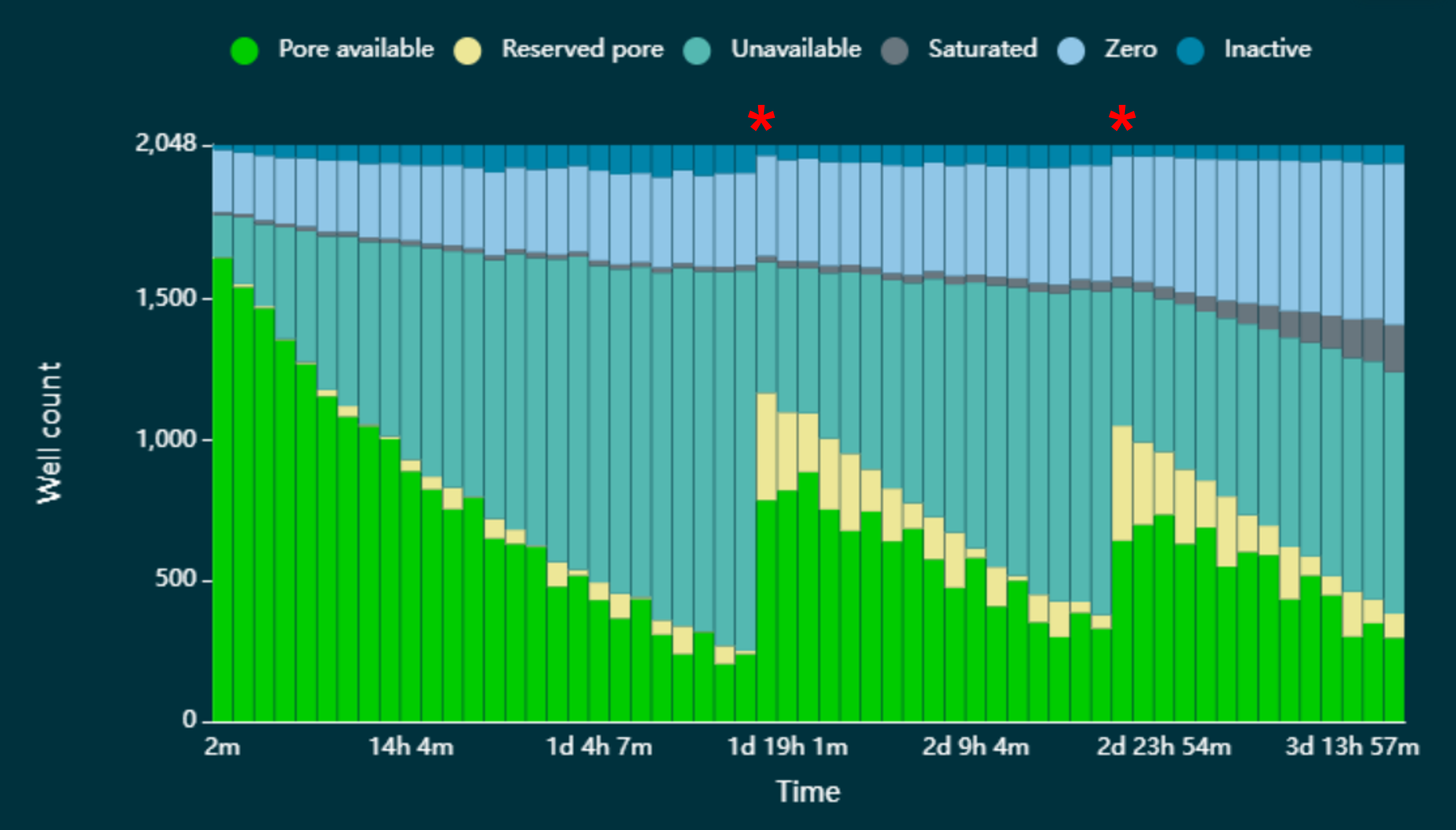
Figure 1. A flow cell has been loaded with a DNA library that has accumulated pores in the 'unavailable' state. The red asterisks indicate when sequencing has been paused and a wash step has been performed. After a wash step was completed, a significant number of pores reverted to the 'single pore' state and were available for sequencing.- This protocol assumes that the flow cells have already had a DNA/RNA library run on them
- The aim is to remove most of the initial library and prepare the flow cells for the loading of a subsequent library
- The Wash Kit contains all solutions required for the removal of the initial library
- Data acquisition in MinKNOW should be stopped (if loading a new library or storing the flow cells), or paused (if loading more of the same library after the wash) during the wash procedure and also during subsequent library addition
- After the flow cells have been washed, a new library can be loaded, or the flow cells can be prepared for storage
-
Place the tube(s) of Wash Mix (WMX) on ice. Do not vortex the tube.
-
Thaw the tube(s) of Wash Diluent (DIL) at room temperature.
-
Mix the contents of Wash Diluent (DIL) thoroughly by vortexing, then spin down briefly and place on ice.
-
Prepare the Wash Mix in a suitable vial for the number of flow cells to wash. Mix well by pipetting, and place on ice. Do not vortex the tube.
Reagent Volume per flow cell Volume X24 flow cells, including excess Volume X48 flow cells, including excess Wash Mix (WMX) 2 μl 52 μl 100 μl Wash Diluent (DIL) 398 μl 10,348 μl 19,900 μl Total 400 μl 10,400 μl 20,000 μl -
Stop or pause the sequencing run in MinKNOW for all flow cells to be washed. Leave the flow cells in the device.
-
Remove the waste buffer, as follows:
- Ensure the inlet port is closed.
- Insert a P1000 pipette into a waste port and remove the waste buffer
Note: As the inlet port is closed, no fluid should leave the sensor array area.
-
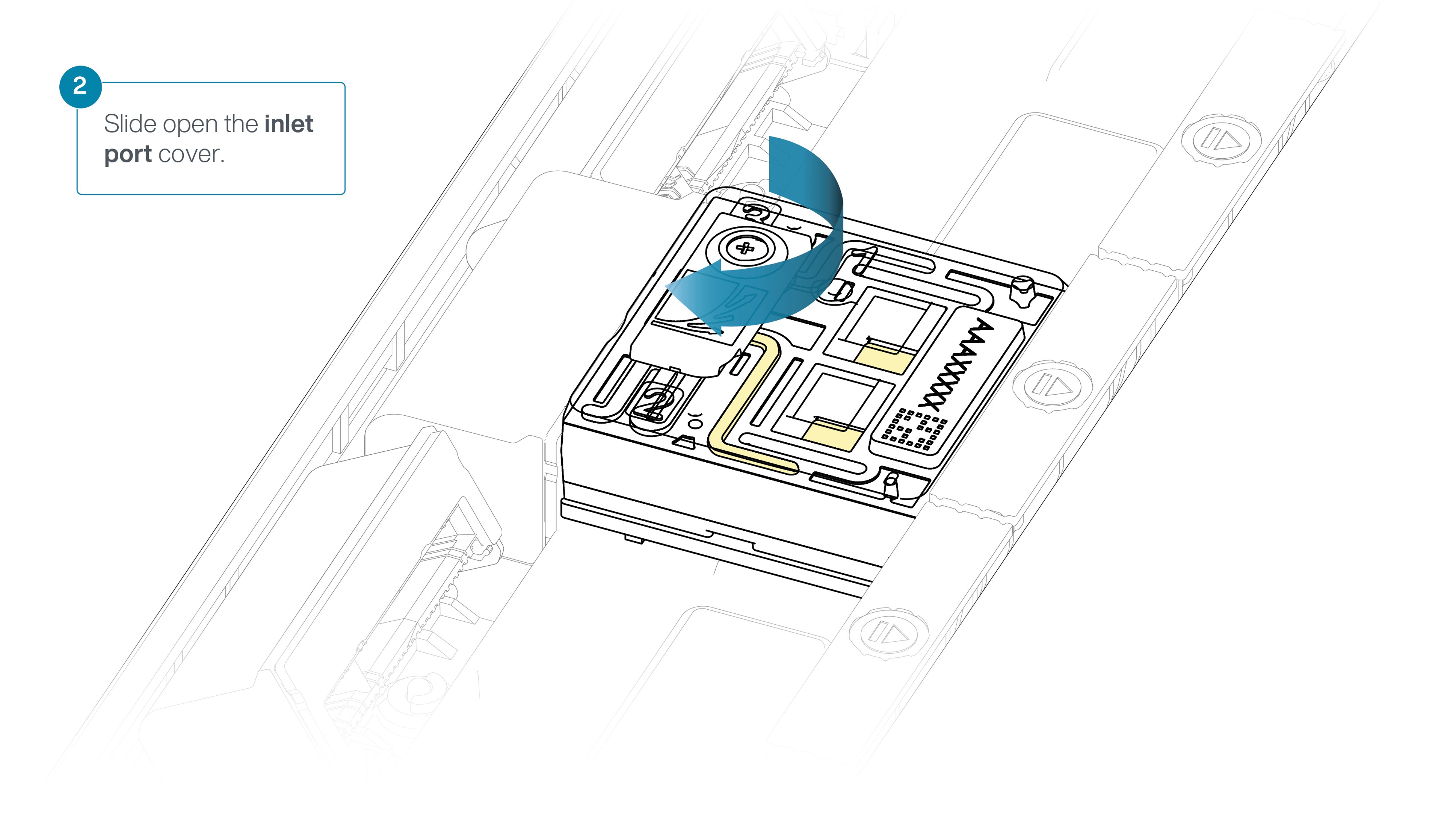
Slide the inlet port cover clockwise to open the inlet port of the first flow cell inserted. Repeat for all flow cells.
-
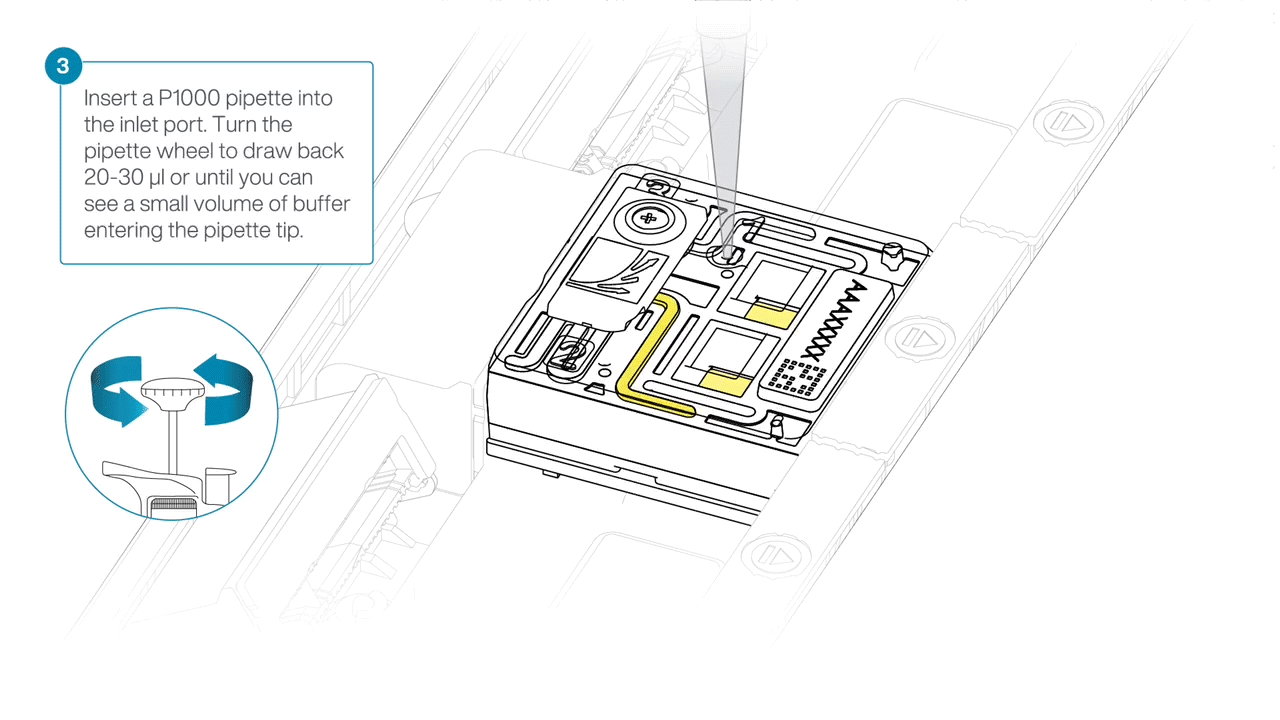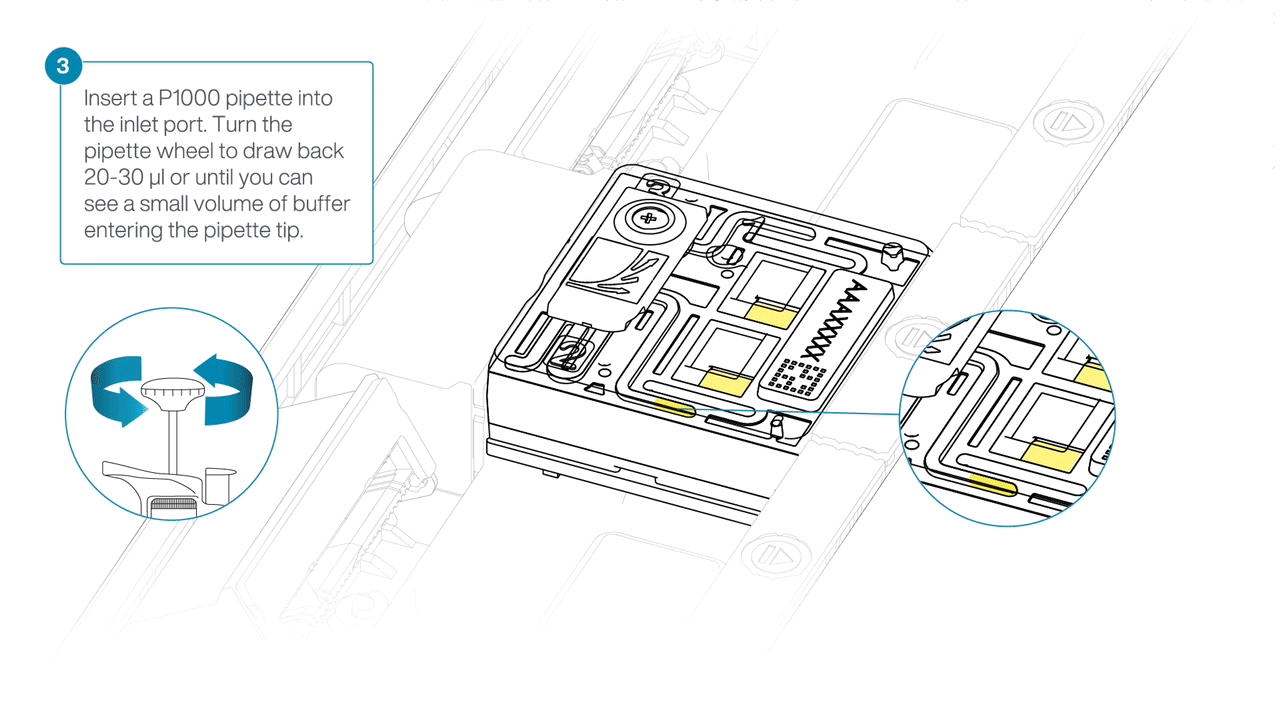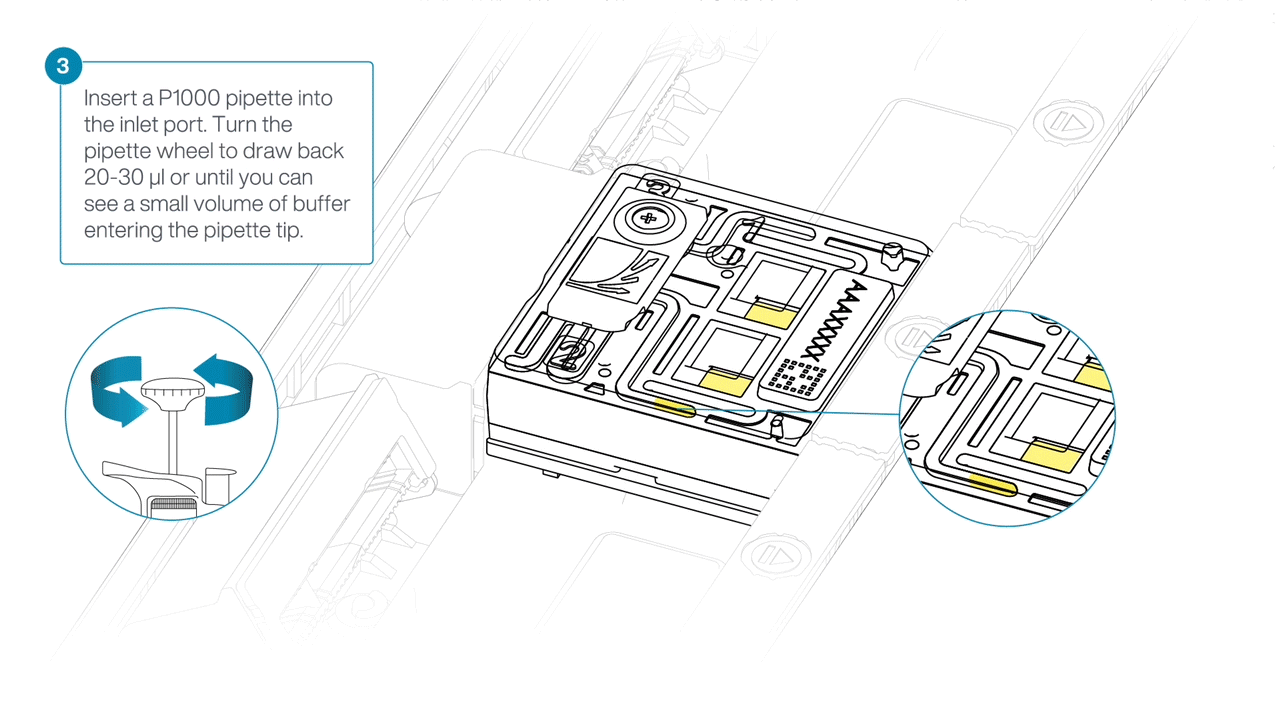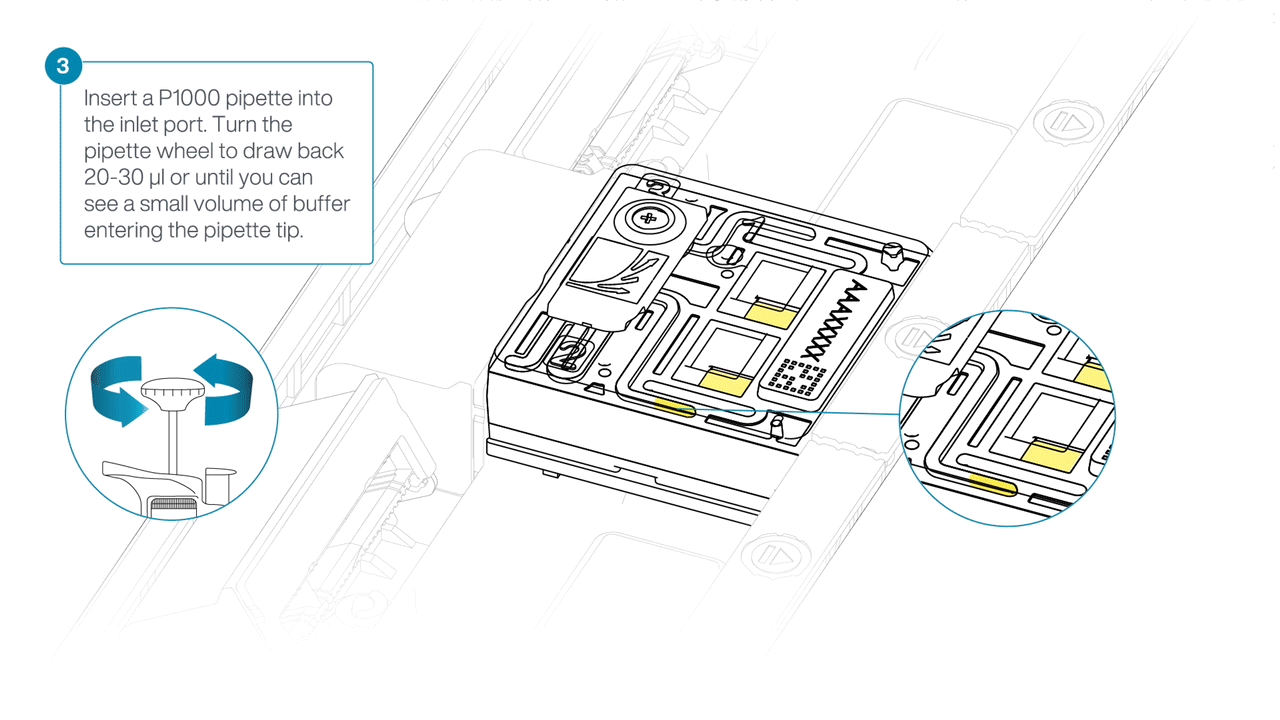
After opening the inlet port, draw back a small volume to remove any air bubbles:
- Set a P1000 pipette to 200 µl.
- For the first flow cell, insert the tip into the inlet port.
- Turn the wheel until the dial shows 220-230 µl, or until you can see a small volume of buffer/liquid entering the pipette tip.
- Repeat for the remaining flow cells using a new pipette tip.
-
Slowly load 200 µl of the prepared flow cell wash mix into the inlet port, as follows:
- Using a P1000 pipette, take 200 µl of the flow cell wash mix
- Insert the pipette tip into the inlet port, ensuring there are no bubbles in the tip
- Slowly twist the pipette wheel down to load the flow cell (if possible with your pipette) or push down the plunger very slowly, leaving a small volume of buffer in the pipette tip.
- Set a timer for a 5 minute incubation.
-
Once the 5 minute incubation time is complete, carefully load the remaining 200 µl of the prepared flow cell wash mix into the inlet port, as follows:
- Using a P1000 pipette, take 200 µl of the flow cell wash mix
- Insert the pipette tip into the inlet port, ensuring there are no bubbles in the tip
- Slowly twist the pipette wheel down to load the flow cell (if possible with your pipette) or push down the plunger very slowly, leaving a small volume of buffer in the pipette tip.
-
Close the inlet port and start a 60 minute timer once the first flow cell has been loaded.
-
Repeat the two-step addition of the wash mix with the 5 minute break in between and the hour incubation for all of the flow cells.
-
After each row (8 flow cells), thoroughly mix the Wash Mix.
-
For each flow cell, remove the waste buffer as follows:
- Ensure the inlet port is closed.
- Insert a P1000 pipette into a waste port and remove the waste buffer
Note: As both the inlet port is closed, no fluid should leave the sensor array area.



