- Materials
-
- 3 µg high molecular weight human genomic DNA
- Sequence capture kit (e.g. Agilent SureSelect Human All Exon, Cat# 232866)
- Custom primer mix, 10 μM (IDT) - see below for sequences
- Ligation Sequencing Kit (SQK-LSK109)
- Flow Cell Priming Kit (EXP-FLP002)
- PCR Expansion (EXP-PCA001)
- Consumables
-
- NEBNext dsDNA Fragmentase (M0348L)
- NEB Blunt/TA Ligase Master Mix (NEB, cat # M0367)
- NEBNext Ultra II End repair/dA-tailing Module (NEB, E7546)
- NEBNext Quick Ligation Module (NEB, E6056)
- Agencourt AMPure XP beads (Beckman Coulter™ cat # A63881)
- Cot-1 DNA (ThermoFisher Scientific 15279-011)
- Dynabeads MyOne Streptavidin T1 (ThermoFisher Scientific, 65601)
- Blocking oligo at 1 mM, sequence 5'-AGGTTAAACACCCAAGCAGACGCCGCAATATCAGCACCAACAGAAACAACC-3'
- LongAmp Taq 2X Master Mix (e.g. NEB, cat # M0287)
- Nuclease-free water (e.g. ThermoFisher, AM9937)
- Freshly prepared 80% ethanol in nuclease-free water
- Freshly prepared 70% ethanol in nuclease-free water
- 0.5 M EDTA, pH 8 (Thermo Scientific, R1021)
- 1.5 ml Eppendorf DNA LoBind tubes
- 0.2 ml thin-walled PCR tubes
- 0.2 ml 96 well PCR plate
- Equipment
-
- Hula mixer (gentle rotator mixer)
- Magnetic rack, suitable for 1.5 ml Eppendorf tubes
- Microfuge
- Vortex mixer
- Thermal cycler
- Ice bucket with ice
- Timer
- SpeedVac
- P1000 pipette and tips
- P200 pipette and tips
- P100 pipette and tips
- P20 pipette and tips
- P10 pipette and tips
- P2 pipette and tips
- Optional equipment
-
- Standard gel electrophoresis equipment
- Agilent Bioanalyzer (or equivalent)
- Qubit fluorometer (or equivalent for QC check)
- Eppendorf 5424 centrifuge (or equivalent)
-
For this protocol, you will need 3 µg high molecular weight human genomic DNA.
-
Input DNA
How to QC your input DNA
It is important that the input DNA meets the quantity and quality requirements. Using too little or too much DNA, or DNA of poor quality (e.g. highly fragmented or containing RNA or chemical contaminants) can affect your library preparation.
For instructions on how to perform quality control of your DNA sample, please read the Input DNA/RNA QC protocol.
Chemical contaminants
Depending on how the DNA is extracted from the raw sample, certain chemical contaminants may remain in the purified DNA, which can affect library preparation efficiency and sequencing quality. Read more about contaminants on the Contaminants page of the Community.
-
Custom primer mix sequences
Please order these sequences at 10 μM from IDT:
Forward primer: 5’ CAATTCGGTCTCCAGTGACTTGCCTGTCGCTCTATCTTC 3’
Reverse Primer: 5’ CAATTCGGTCTCCCACTTTTCTGTTGGTGCTGATATTGC 3’ -
Ligation Sequencing Kit contents (SQK-LSK109)
Name Acronym Cap colour No. of vials Fill volume per vial (µl) DNA CS DCS Yellow 1 50 Adapter Mix AMX Green 1 40 Ligation Buffer LNB Clear 1 200 L Fragment Buffer LFB White cap, orange stripe on label 2 1,800 S Fragment Buffer SFB Grey 2 1,800 Sequencing Buffer SQB Red 2 300 Elution Buffer EB Black 1 200 Loading Beads LB Pink 1 360 -
Flow Cell Priming Kit contents (EXP-FLP002)
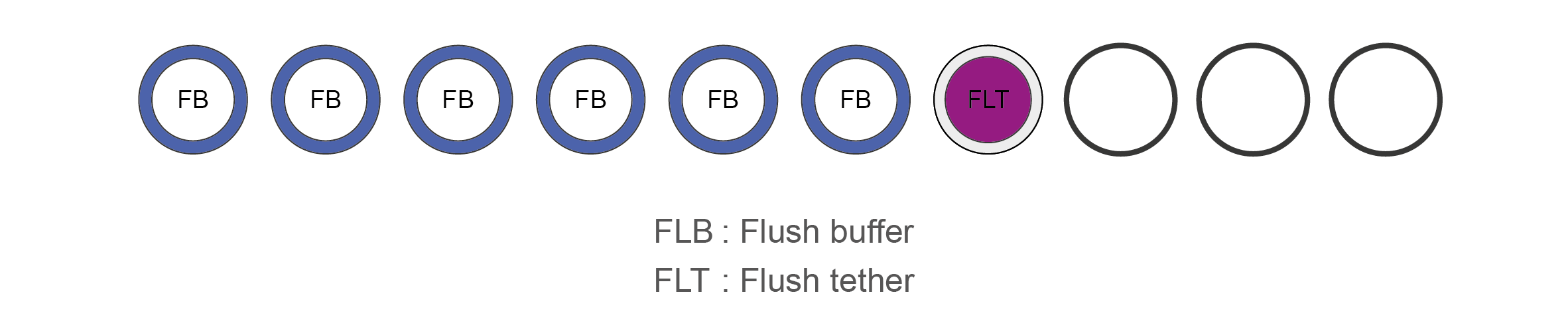
Name Acronym Cap colour No. of vials Fill volume per vial (μl) Flush Buffer FB Blue 6 1,170 Flush Tether FLT Purple 1 200
